Advertisements
Advertisements
प्रश्न
Arrange the following as per instruction given in the bracket.
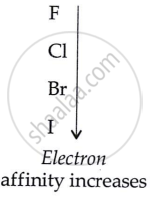
Cl, F, Br, I (increasing electron affinity)
उत्तर
In a group, Electron Affinity increases from top to bottom.
F < Cl < Br < I
संबंधित प्रश्न
The energy released when an electron is added to a neutral gaseous isolated atom to form a negatively charged iron.
Define the term ‘electron affinity’.
Arrange the elements of second period in increasing order of their electron affinity. Name the elements which do not follow the trend in this period.
An element in period 3, whose electron affinity is zero:
A, B, C are three elements in which B is an inert gas other than helium.With this information complete the following table.
| Element | Atomic number | No. of electrons in the valence shell | Group to which the element belongs |
| A | Z - 1 | ||
| B | Z | ||
| C | Z + 1 |
Also, explain the following : Electron affinity of element A is more than that that of element C.
Fill in the blank
The nature of oxide Al2O3 is ____
The following questions refer to the Periodic Table.
Which of the elements has the greatest electron affinity among the halogens?
Arrange the following as per instruction given in the bracket.
Cl, F, Br, I (increasing electron affinity)
The electron affinity of an element X is greater than that of element Y.
How is the electronegativity of X likely to compare with that of Y?
Among period 2 elements A, B, C and D, the one which has highest electron affinity is ______.
