Advertisements
Advertisements
Question
Arrange the following as per instruction given in the bracket.
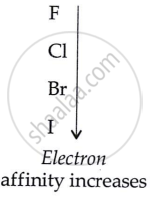
Cl, F, Br, I (increasing electron affinity)
Solution
In a group, Electron Affinity increases from top to bottom.
F < Cl < Br < I
RELATED QUESTIONS
Draw an electron dot diagram to show the formation of each of the following compounds:
Magnesium Chloride
[H = 1, C = 6, Mg = 12, Cl = 17]
Element X is a metal with a valency 2, Y is 3 non- metal with a valency 3.
1) Write an equation to show how Y from an ion.
2) If Y is a diatomic gas, write an equation for the direct combination of X and Y to form a compound.
Give one word or phrase for the following:
The amount of energy released when an atom in the gaseous state accepts an electron to form an anion.
Define the term ‘electron affinity’.
Which has higher E.A. fluorine or Neon?
Why is the electron affinity of fluorine less than chlorine?
A, B, C are three elements in which B is an inert gas other than helium.With this information complete the following table.
| Element | Atomic number | No. of electrons in the valence shell | Group to which the element belongs |
| A | Z - 1 | ||
| B | Z | ||
| C | Z + 1 |
Also, explain the following : Electron affinity of element A is more than that that of element C.
State whether the following statement is true or false
The elements with higher electron affinity have higher ionization potential.
Explain
Halogens have high electron affinity.
With reference to the variation of properties in the Periodic Table, which of the following is generally true?
Electron affinity increases going down a group.
