Advertisements
Advertisements
प्रश्न
Arrange the following.
In decreasing order of basic strength
![]()
उत्तर
Aliphatic amines are more basic than aromatic amines. Therefore CH3CH2NH2 and CH3NH2 are more basic. Among the ethylamine and methylamine. ethylamine was experienced more +I effect than methylamine and hence ethylamine is more basic than methylamine.
Nitro group has a powerful electron-withdrawing group and they have both – R effect as well as – I effect. As a result, all the nitro anilines are weaker bases than aniline. In P-nitro aniline
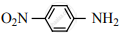
both – R effect and – I effect of the NO2 group decrease the basicity. Therefore decreasing order of basic strength is,
![]()
Ethylamine > Methylamine > Aniline > p-nitro aniline
APPEARS IN
संबंधित प्रश्न
In the following
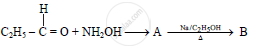
The compound ‘B’ is _______.
(A) Propan–1–amine
(B) Propan–2–amine
(C) Isopropylamine
(D) Dimethylamine
Choose the most correct option.
Carbylamine test is given by ____________.
Choose the most correct option.
Identify ‘B’ in the following reactions
\[\ce{CH3 - C ≡ N ->[Na/C2H5OH] A ->[NaNO2/dilHCl]B}\]
A tertiary amine is an organic compound ____________.
How will you convert nitrobenzene into m-nitro aniline?
How will you prepare propan-1-amine from propanamide?
Write short note on the following.
Ammonolysis
Define Amines.
Write short note on the following.
Ammonolysis
Write short note on Ammonolysis.
