Advertisements
Advertisements
प्रश्न
Arrange the following in decreasing order of their acidic strength and give reason for your answer.
\[\ce{CH3CH2OH, CH3COOH, ClCH2COOH, FCH2COOH, C6H5CH2COOH}\]
उत्तर
The order is
\[\ce{ClCH2COOH > FCH2COOH > C6H5CH2COOH > CH3COOH > CH3CH2OH }\]
When compared to other alcohols \[\ce{CH3CH2OH}\] is the least acidic. Because of the halogen, \[\ce{FCH2COOH}\] is quite acidic. It is either an electron-donating group or an electron receiving group in acidic strength. Acidic strength increases as the number of electron withdrawing groups increases. The acidic strength of electron-donating groups diminishes.
APPEARS IN
संबंधित प्रश्न
Write the structures and IUPAC names of the α - methyl butyraldehyde.
Name the following compound according to IUPAC system of nomenclature:
CH3CH2COCH(C2H5)CH2CH2Cl
Name the following compound according to IUPAC system of nomenclature:
CH3COCH2COCH3
Name the following compound according to IUPAC system of nomenclature:
CH3CH(CH3)CH2C(CH3)2COCH3
Name the following compound according to IUPAC system of nomenclature:
(CH3)3CCH2COOH
Give IUPAC name of :
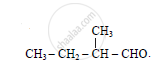
Give the IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{.................}\ce{Br}\phantom{....}\ce{CH3}\phantom{.........}\ce{O}\phantom{...}\\
\phantom{................}|\phantom{......}|\phantom{............}||\phantom{..}\\
\ce{CH3 - CH2 - CH2CH - CH - CH2 - C - H}
\end{array}\]
Give IUPAC names of the following compound:
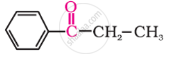
The following compound is called:
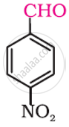
Write the structure of the following compound:
Di-sec. butyl ketone
