Advertisements
Advertisements
प्रश्न
Assertion: Amimonolysis of alkyl halides involves the reaction between alkyl halides and alcoholic ammonia.
Reason: Ammonolysis of alkyl halides produces secondary amines only.
विकल्प
Both Assertion and Reason are true and Reason is the correct explanation for Assertion.
Both Assertion and Reason are true but Reason is not the correct explanation for Assertion.
Assertion is true but Reason is false.
Assertion is false but Reason is true.
उत्तर
Assertion is true but Reason is false.
Explanation:
When an alkyl or benzyl halide combines with an ethanolic solution of ammonia, an amino (-NH2) group replaces the halogen atom via a nucleophilic substitution reaction. Ammonolysis continues until all of the hydrogen in ammonia has been replaced. As a result, a combination of primary, secondary and tertiary amines are produced. Thus, the assertion is correct, but the reasoning is incorrect.
APPEARS IN
संबंधित प्रश्न
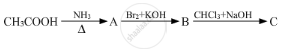
Give the structures of A, B and C in the following reactions :
Which of the following reactions is appropriate for converting benzamide to aniline?
Best method for preparing primary amines from alkyl halides without changing the number of carbon atoms in the chain is ______.
The reagents that can be used to convert benzenediazonium chloride to benzene are:
(i) \[\ce{SnCl2/HCl}\]
(ii) \[\ce{CH3CH2OH}\]
(iii) \[\ce{H3PO2}\]
(iv) \[\ce{LiAlH4}\]
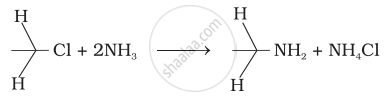
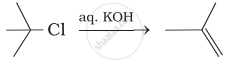
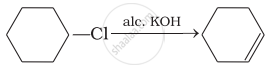
Which of the following reactions are correct?
(i)
(ii)
(iii)
(iv)
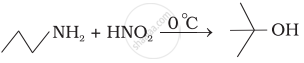
Describe Gabriel's phthalimide synthesis. (Give reaction)
C6H5CONHCH3 can be converted into C6H5CH2NHCH3 by:-
When primary amines are treated with HCl, the product obtained is which of the following?
Which of the following reaction DOES NOT involve Hoffmann bromamide degradation?
Identify the product ‘C’ in the following reaction.
\[\ce{Aniline ->[(CH3CH)2O][Pyridine] A ->[Br2][CH3COOH] B ->[H^+ or OH^-] C}\]
