Advertisements
Advertisements
Question
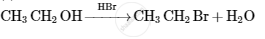
Assertion: Amimonolysis of alkyl halides involves the reaction between alkyl halides and alcoholic ammonia.
Reason: Ammonolysis of alkyl halides produces secondary amines only.
Options
Both Assertion and Reason are true and Reason is the correct explanation for Assertion.
Both Assertion and Reason are true but Reason is not the correct explanation for Assertion.
Assertion is true but Reason is false.
Assertion is false but Reason is true.
Solution
Assertion is true but Reason is false.
Explanation:
When an alkyl or benzyl halide combines with an ethanolic solution of ammonia, an amino (-NH2) group replaces the halogen atom via a nucleophilic substitution reaction. Ammonolysis continues until all of the hydrogen in ammonia has been replaced. As a result, a combination of primary, secondary and tertiary amines are produced. Thus, the assertion is correct, but the reasoning is incorrect.
APPEARS IN
RELATED QUESTIONS
Write the chemical equation involved in the following reaction:
Hoffmann-bromamide degradation reaction
Accomplish the following conversion:
Benzamide to toluene
Identify 'A' and 'B' in the following reaction and rewrite the complete reaction :
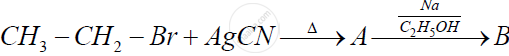
Give the structures of A, B and C in the following reactions :
Identify the product obtained, when benzamide is treated with bromine and aqueous sodium hydroxide.
Identify the INCORRECT statement regarding Hofmann bromamide reaction.
Reduction of aromatic nitro compounds using \[\ce{Fe}\] and \[\ce{HCl}\] gives ______.
Write following conversions:
nitrobenzene `->` acetanilide
Reduction of nitro alkanes yields which compound?
Acetamide and ethyl amide can be distinguished by reacting with.
