Advertisements
Advertisements
प्रश्न
‘Atoms of the same element or different from Dalton’s atomic theory by the Modern Atomic Theory.
Give two examples each of –
- Atoms of the same element
- Atoms of different elements combining to form a molecule.
उत्तर
Atoms of the same element or different elements combine to form a molecule.
Atoms of different elements forming a molecule
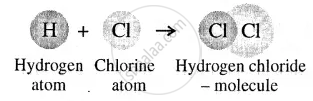
A molecule is the smallest particle of a pure substance – element or compound
- which can exist independently and
- retain the physical & chemical properties of the substance.
| Elements of same kind | Molecule of elements |
| Hydrogen atom |
Hydrogen molecule
|
| Nitrogen atom |
Nitrogen molecule
|
| Elements of same kind | Molecule of elements |
| Hydrogen [2 atoms] | 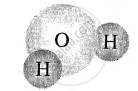 |
| Oxygen [1 atom] | water molecule |
APPEARS IN
संबंधित प्रश्न
Write the molecular formulae of Sodiun sulphide:
Write the molecular formulae of Calcium hydroxide :
The valency of sodium is one, write the molecular formula for sodium sulphate ?
The valency of sodium is one, write the molecular formula for sodium carbonate ?
Fill in the blank
_______ is a triatomic molecule.
State the term
The symbolic representation of a molecule ______
What is a molecule ? Explain with an example.
Write the cations and anions present, if any, in the following :
- CH3COONa
- NaCl
- H2
- NH4NO3
Fill in the following blanks :
1 mole contains .............. atoms, molecules or ions of a substance.
If one mole of nitrogen molecules weighs 28 g, calculate the mass of one molecule of nitrogen in grams.
Which contains more molecules, 10 g of sulphur dioxide (SO2) or 10 g of oxygen (O2) ?
(Atomic masses : S = 32 u ; O = 16 u)
What is the number of electrons after the transfer of electrons in magnesium and sulphur atoms.
‘Dilute hydrochloric acid is added to sodium hydroxide’. Give a word equation and a molecular equation of the same. Is the equation written, a balanced equation. Give reasons.
Fill in the blank:
A molecule of an element is _________.
Fill in the blank:
The formula of aluminium sulphate is _________.
What is the fraction of the mass of water due to neutrons?
Express each of the following in kilograms
5.84 × 10−3 mg
Express each of the following in kilograms
58.34 g
______ of an element consist of a fixed number of different types of atom.


