Advertisements
Advertisements
प्रश्न
‘Atoms of the same element or different from Dalton’s atomic theory by the Modern Atomic Theory.
Give two examples each of –
- Atoms of the same element
- Atoms of different elements combining to form a molecule.
उत्तर
Atoms of the same element or different elements combine to form a molecule.
Atoms of different elements forming a molecule
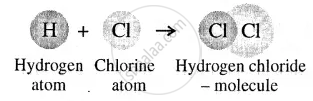
A molecule is the smallest particle of a pure substance – element or compound
- which can exist independently and
- retain the physical & chemical properties of the substance.
| Elements of same kind | Molecule of elements |
| Hydrogen atom |
Hydrogen molecule
|
| Nitrogen atom |
Nitrogen molecule
|
| Elements of same kind | Molecule of elements |
| Hydrogen [2 atoms] | 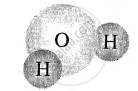 |
| Oxygen [1 atom] | water molecule |
APPEARS IN
संबंधित प्रश्न
What does 6NaNO3 represent ?
Write the molecular formulae of Sodiun sulphide:
Write the molecular formulae of Iron (III) sulphide:
Write the molecular formulae of Nitric acide :
The valency of sodium is one, write the molecular formula for sodium oxide ?
The valency of sodium is one, write the molecular formula for sodium carbonate ?
What is a molecule ? Explain with an example.
In hydrogen peroxide (H2O2), the proportion of hydrogen and oxygen by mass is :
Calculate the number of molecules present in a drop of chloroform (CHCl3) weighing 0.0239 g.
(Atomic masses : C = 12 u ; H = 1 u ; Cl = 35.5 u)
If 1 gram of sulphur dioxide contains x molecules, how many molecules will be present in 1 gram of oxygen ?
(S = 32 u ; O = 16 u)
Diagrammatically represent the transfer of electrons.
Explain the term ‘atomicity’.
Fill in the blank:
A molecule of an element is _________.
State true of false. If false, give the correct statement.
Atoms cannot exist alone. They can only exist as groups called molecules.
Define molecule.
A group of atoms carrying a fixed charge on them is called ————.
______ can be formed by the same or different kinds of atoms.


