Advertisements
Advertisements
प्रश्न
Balance the following chemical equation and identify the type of chemical reaction.
`"HgO"("s") overset("(heat)")(->) "Hg"("l") + "O"_2("g")`
उत्तर
The chemical equation in balanced form may be written as follows :
`2"HgO"("s") overset("(heat)")(->) 2"Hg"("l") + "O"_2("g")` - Decomposition reaction
APPEARS IN
संबंधित प्रश्न
Rewrite the following statements by selecting the correct options:
\[\ce{CaCO3 ->[\Delta] CaO + CO2 ^}\] is a __________ reaction.
A solid substance P which is very hard is used in the construction of many buildings, especially flooring. When substance P is heated strongly, it decomposes to form another solid Q and a gas R is given out. Solid Q reacts with water with the release of a lot of heat to form a substance S. When gas R is passed into a clear solution of substance S, then a white precipitate of substance T is formed. The substance T has the same chemical composition as starting substance P.
(a) What is substance P? Write its common name as well as chemical formula.
(b) What is substance Q?
(c) What is gas R?
(d) What is substance S? What is its clear solution known as?
(e) What is substance T? Name any two natural forms in which substance T occurs in nature.
Give one example of a decomposition reaction which is carried out with electricity.
What type of reaction is represented by the following equation?
2 FeSO4 → Fe2 O3 + SO2 + SO3
Which of the following can be decomposed by the action of light?
(a) NaCl
(b) KCl
(c) AgCl
(d) CuCl
What do you mean by redox reaction ? Explain with the help of an example.
Classify the following reaction into different type:
\[\ce{2KClO3(s)->[\Delta] 2KCl(aq) + 3O2(g)}\]
Assertion: Decomposition of vegetable matter into compost is an endothermic reaction.
Reason: Decomposition reaction involves breakdown of a single reactant into simpler products.
Marble statues are corroded or stained rain water. Identify the main reason.
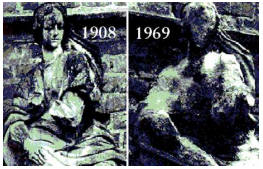
Write one equation for decomposition reactions where energy is supplied in the form of light.
