Advertisements
Advertisements
प्रश्न
Write one equation for decomposition reactions where energy is supplied in the form of light.
उत्तर
Decomposition reaction involving absorption of light,
- \[\ce{\ce{\underset{Hydrogen peroxide}{2H2O2_{(l)}}}->[Light] \underset{Water}{2H2O_{(l)}} + \underset{Oxygen}{O2_{(g)}}}\]
- \[\ce{2AgCl_{(s)} ->[sunlight] 2Ag_{(s)} + Cl2_{(g)}}\]
संबंधित प्रश्न
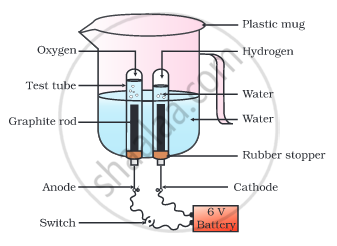
Why is the amount of gas collected in one of the test tubes in the following Activity double of the amount collected in the other? Name this gas.
- Take a plastic mug. Drill two holes at its base and fit rubber stoppers in these holes. Insert carbon electrodes in these rubber stoppers as shown in the following Fig.
- Connect these electrodes to a 6 volt battery.
- Fill the mug with water such that the electrodes are immersed. Add a few drops of dilute sulphuric acid to the water.
- Take two test tubes filled with water and invert them over the two carbon electrodes.
- Switch on the current and leave the apparatus undisturbed for some time.
- You will observe the formation of bubbles at both the electrodes. These bubbles displace water in the test tubes.
- Is the volume of the gas collected the same in both the test tubes?
• Once the test tubes are filled with the respective gases, remove them carefully. - Test these gases one by one by bringing a burning candle close to the mouth of the test tubes.
Caution: This step must be performed carefully by the teacher.
- What happens in each case?
- Which gas is present in each test tube?
What type of reaction is represented by the following equation?
2 FeSO4 → Fe2 O3 + SO2 + SO3
What type of chemical reaction take place when lime-stone is heated?
What type of chemical reaction take place when electricity is passed through water?
What type of chemical reaction is represented by the following equation?
X → Y + Z
What type of reaction is represented by the following equation?
NH4NO2 → N2 + 2H2O
Which of the following can be decomposed by the action of light?
(a) NaCl
(b) KCl
(c) AgCl
(d) CuCl
Classify the following reaction as combination, decomposition, displacement, precipitation and neutralization. Also balance the equation.
\[\ce{Zn_{(s)} + H2SO4 -> ZnSO4_{(s)} + H2_{(g)}}\]
Explain the following type of chemical reaction, giving two examples for it:
Decomposition reaction
MULTIPLE CHOICE QUESTIONS
Tick the most appropriate answer.
Thermal decomposition of a substance is brought about with the help of
- reactants
- water
- wind
- heat
What do you mean by redox reaction ? Explain with the help of an example.
What does one mean by endothermic reaction? Give example.
Identify the type of following reaction :
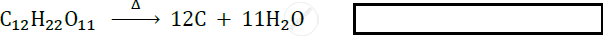
(a) Design an activity to demonstrate the decomposition reaction of lead nitrate.
(b) Draw labelled diagram of the experimental set-up. List two main observations.
(c) Write balanced chemical equation for the reaction stating the physical state of the reactant and the products.
Answer the following question.
2 g of silver chloride is taken in a china dish and the china dish is placed in sunlight for some time. What will be your observation in this case? Write the chemical reaction involved in the form of a balanced chemical equation. Identify the type of chemical reaction.
Give a balanced equation for the following type of reaction:
A thermal decomposition reaction in which a metallic nitrate decomposes to give – a basic oxide.
Which one of the following processes involves chemical reactions?
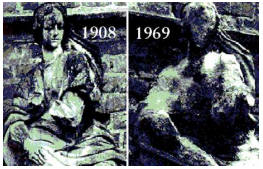
Marble statues are corroded or stained rain water. Identify the main reason.
Balance the following chemical equation and identify the type of chemical reaction.
`"HgO"("s") overset("(heat)")(->) "Hg"("l") + "O"_2("g")`
Write one equation for decomposition reactions where energy is supplied in the form of electricity.
