Advertisements
Advertisements
प्रश्न
What type of reaction is represented by the following equation?
NH4NO2 → N2 + 2H2O
उत्तर
Decomposition reaction
APPEARS IN
संबंधित प्रश्न
Taking into consideration the relationship in the first pair, complete the second pair
2H2 + O2 → 2H2O :Combination Reaction :: 2HgO → 2Hg + O2:_________
A solid substance P which is very hard is used in the construction of many buildings, especially flooring. When substance P is heated strongly, it decomposes to form another solid Q and a gas R is given out. Solid Q reacts with water with the release of a lot of heat to form a substance S. When gas R is passed into a clear solution of substance S, then a white precipitate of substance T is formed. The substance T has the same chemical composition as starting substance P.
(a) What is substance P? Write its common name as well as chemical formula.
(b) What is substance Q?
(c) What is gas R?
(d) What is substance S? What is its clear solution known as?
(e) What is substance T? Name any two natural forms in which substance T occurs in nature.
Give one example of a decomposition reaction which is carried out by applying heat.
What type of reaction is represented by the following equation?
2 FeSO4 → Fe2 O3 + SO2 + SO3
What is the colour of ferrous sulphate crystals? How does this colour change after heating?
What is a decomposition reaction?
What are thermal decomposition reactions ? Explain with an example.
What do you mean by redox reaction ? Explain with the help of an example.
Identify the type of following reaction :
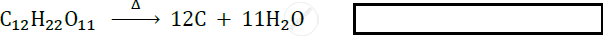
Differentiate between direct combination reaction and a decomposition reaction.
