Advertisements
Advertisements
प्रश्न
Give one example of a decomposition reaction which is carried out by applying heat.
उत्तर
Example of the decomposition reaction are as follows:
by applying heat:
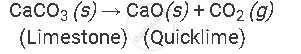
APPEARS IN
संबंधित प्रश्न
What type of reaction is represented by the following equation?
NH4NO2 → N2 + 2H2O
Name the product formed on strongly heating ferrous sulphate crystals. What type of chemical reaction occurs in this change?
Which of the following can be decomposed by the action of light?
(a) NaCl
(b) KCl
(c) AgCl
(d) CuCl
Classify the following reaction as combination, decomposition, displacement, precipitation and neutralization. Also balance the equation.
\[\ce{Zn_{(s)} + H2SO4 -> ZnSO4_{(s)} + H2_{(g)}}\]
A student wants to study a decomposition reaction by taking ferrous sulphate crystals. Write two precautions he must observe while performing the experiment.
Give a balanced equation for –
A thermal decomposition reaction involving heat on limestone [calcium carbonate]
Differentiate between the following:
Thermal decomposition and thermal dissociation.
What is electrolysis?
Explain the reaction given in the figure.

Which of the following is an endothermic process?
