Advertisements
Advertisements
प्रश्न
Balance the following reaction by oxidation number method.
\[\ce{Bi(OH)_{3(s)} + Sn(OH)^-_{3(aq)}->Bi_{(s)} + Sn(OH)^2-_{6(aq)}(basic)}\]
उत्तर
\[\ce{Bi(OH)_{3(s)} + Sn(OH)^-_{3(aq)}->Bi_{(s)} + Sn(OH)^2-_{6(aq)}(basic)}\]
Step 1: Write the skeletal equation and balance the elements other than O and H.
\[\ce{Bi(OH)_{3(s)} + Sn(OH)^-_{3(aq)}->Bi_{(s)} + Sn(OH)^2-_{6(aq)}}\]
Step 2: Assign oxidation numbers to Bi and Sn. Calculate the increase and decrease in the oxidation number and make them equal.
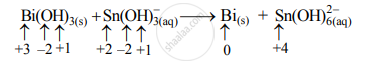
Increase in oxidation number:
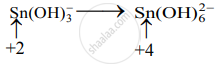
(Increase per atom = 2)
Decrease in oxidation number:

(Decrease per atom = 3)
To make the net increase and decrease equal, we must take 3 atoms of Sn and 2 atoms of Bi.
\[\ce{2Bi(OH)_{3(s)} + 3Sn(OH)^-_{3(aq)}->2Bi_{(s)} + 3Sn(OH)^2-_{6(aq)}}\]
Step 3: Balance ‘O’ atoms by adding 3H2O to the left-hand side.
\[\ce{2Bi(OH)_{3(s)} + 3Sn(OH)^-_{3(aq)} + 3H2O_{(l)}->2Bi_{(s)} + 3Sn(OH)^2-_{6(aq)}}\]
Step 4: The medium is basic. To make hydrogen atoms on the two sides equal, add 3H+ on the right-hand side.
\[\ce{2Bi(OH)_{3(s)} + 3Sn(OH)^-_{3(aq)} + 3H2O_{(l)}->2Bi_{(s)} + 3Sn(OH)^2-_{6(aq)} + 3H^+_{( aq)}}\]
Add OH− ions equal to the number of H+ ions on both sides of the equation.
\[\ce{2Bi(OH)_{3(s)} + 3Sn(OH)^-_{3(aq)} + 3H2O_{(l)} + 3OH^-_{( aq)}->2Bi_{(s)} + 3Sn(OH)^2-_{6(aq)} + 3H^+_{( aq)} + 3OH^-_{( aq)}}\]
The H+ and OH− ions appearing on the same side of the reaction are combined to give H2O molecules.
\[\ce{2Bi(OH)_{3(s)} + 3Sn(OH)^-_{3(aq)} + 3H2O_{(l)} + 3OH^-_{( aq)}->4Bi_{(s)} + 3Sn(OH)^2-_{6(aq)} + 3H2O_{(l)}}\]
\[\ce{2Bi(OH)_{3(s)} + 3Sn(OH)^-_{3(aq)} + 3OH^-_{( aq)}->2Bi_{(s)} + 3Sn(OH)^2-_{6(aq)}}\]
Step 5: Check two sides for balance of atoms and charges.
Hence, balanced equation: \[\ce{2Bi(OH)_{3(s)} + 3Sn(OH)^-_{3(aq)} + 3OH^-_{( aq)}->2Bi_{(s)} + 3Sn(OH)^2-_{6(aq)}}\]
APPEARS IN
संबंधित प्रश्न
Consider the reaction:
\[\ce{O3(g) + H2O2(l) → H2O(l) + 2O2(g)}\]
Why it is more appropriate to write these reaction as:
\[\ce{O3(g) + H2O2 (l) → H2O(l) + O2(g) + O2(g)}\]
Also, suggest a technique to investigate the path of the redox reactions.
The compound AgF2 is an unstable compound. However, if formed, the compound acts as a very strong oxidizing agent. Why?
Balance the following equation in the basic medium by ion-electron method and oxidation number methods and identify the oxidising agent and the reducing agent.
\[\ce{N2H4(l) + ClO^-_3 (aq) → NO(g) + Cl–(g)}\]
Balance the following equation in basic medium by ion-electron method and oxidation number methods and identify the oxidising agent and the reducing agent.
\[\ce{Cl_2O_{7(g)} + H_2O_{2(aq)} -> ClO-_{2(aq)} + O_{2(g)} + H+_{(aq)}}\]
Choose the correct option.
For the following redox reactions, find the correct statement.
\[\ce{Sn^{2⊕} + 2Fe^{3⊕}->Sn^{4⊕} + 2Fe^{2⊕}}\]
Balance the following reaction by oxidation number method.
\[\ce{Cr2O^2-_{7(aq)} + SO^2-_{3(aq)}->Cr^3+_{ (aq)} + SO^2-_{4(aq)}(acidic)}\]
Balance the following reaction by oxidation number method.
\[\ce{H2SO4_{(aq)} + C_{(s)}->CO2_{(g)} + SO2_{(g)} + H2O_{(l)}(acidic)}\]
Which of the following is INCORRECT for the following reaction?
\[\ce{2Zn_{(s)} + O2_{(g)} -> 2ZnO_{(s)}}\]
What is the change in oxidation number of Sulphur in following reaction?
\[\ce{MnO^-_{4(aq)} + SO^{2-}_{3(aq)} -> MnO^{2-}_{4(aq)} + SO^{2-}_{4(aq)}}\]
Identify the oxidising agent in the following reaction:
\[\ce{CH4_{(g)} + 2O2_{(g)} -> CO2_{(g)} + 2H2O_{(l)}}\]
When methane is burnt completely, oxidation state of carbon changes from ______.
Consider the reaction:
\[\ce{6 CO2(g) + 6H2O(l) → C6 H12O6(aq) + 6O2(g)}\]
Why it is more appropriate to write these reaction as:
\[\ce{6CO2(g) + 12H2O(l) → C6 H12O6(aq) + 6H2O(l) + 6O2(g)}\]
Also, suggest a technique to investigate the path of the redox reactions.
Write balanced chemical equation for the following reactions:
Reaction of liquid hydrazine \[\ce{(N2H4)}\] with chlorate ion \[\ce{(ClO^{-}3)}\] in basic medium produces nitric oxide gas and chloride ion in gaseous state.
Write balanced chemical equation for the following reactions:
Dichlorine heptaoxide \[\ce{(Cl2O7)}\] in gaseous state combines with an aqueous solution of hydrogen peroxide in acidic medium to give chlorite ion \[\ce{(ClO^{-}2)}\] and oxygen gas. (Balance by ion-electron method)
Balance the following equations by the oxidation number method.
\[\ce{Fe^{2+} + H^{+} + Cr2O^{2-}7 -> Cr^{3+} + Fe^{3+} + H2O}\]
Balance the following equations by the oxidation number method.
\[\ce{MnO2 + C2O^{2-}4 -> Mn^{2+} + CO2}\]
Identify the redox reactions out of the following reactions and identify the oxidising and reducing agents in them.
\[\ce{3HCl (aq) + HNO3 (aq) -> Cl2 (g) + NOCl (g) + 2H2O (l)}\]
Identify the redox reactions out of the following reactions and identify the oxidising and reducing agents in them.
\[\ce{HgCl2 (aq) + 2KI (aq) -> HgI2 (s) + 2KCl (aq)}\]
Identify the redox reactions out of the following reactions and identify the oxidising and reducing agents in them.
\[\ce{PCl3 (l) + 3H2O (l) -> 3HCl (aq) + H3PO3 (aq)}\]
Balance the following ionic equations.
\[\ce{Cr2O^{2-}7 + Fe^{2+} + H+ -> Cr^{3+} + Fe^{3+} + H2O}\]
Balance the following ionic equations.
\[\ce{MnO^{-}4 + H^{+} + Br^{-} -> Mn^{2+} + Br2 + H2O}\]
In \[\ce{Cu^{2+} + Ag -> Cu + Ag^+}\], oxidation half-reaction is:
In the reaction of oxalate with permanganate in an acidic medium, the number of electrons involved in producing one molecule of CO2 is ______.
