Advertisements
Advertisements
प्रश्न
Calculate the mass of nitrogen supplied to soil by 5 kg of urea [CO(NH2)2].
[O = 16; N = 14; C = 12; H = 1]
उत्तर
Molecular mass of Urea [CO(NH2)2] = [12 + 16 + (14 + 2 × 1) × 2]
= [12 + 16 + 32] = 60
Weight of nitrogen in 60 kg = 28 kg
∴ Weight of nitrogen in 5 kg = `28/0 xx 5`
= 2.33 kg
APPEARS IN
संबंधित प्रश्न
Give balanced chemical equations for the following conversions A, B, and C:
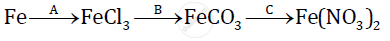
Calculate the percentage of water in ferrous sulphate crystals.
[Fe = 56, S = 32, O =16, H = 1].
What is the mass of nitrogen in 1000Kg of urea [CO(NH2)2] ?
[H = 1, C= 12, N= 14, O = 16]
If the relative molecular mass of A is 90, what is the molecular formula of A?
Water can split into hydrogen and oxygen under suitable conditions. The equations representing the change is: 2H2O(I) → 2H2 (g) + O2(g)
Ammonia burns in oxygen and the combustion in the presence of a catalyst may be represented as:
2NH3 (g) +21/2O2 (g) → 2NO (g) + 3H2O (I)
What mass of steam is produced when 1.5 g of nitrogen monoxide is formed?
The reaction of potassium permanganate (VII) with acidified iron (II) sulphate is given below:
2KMno4 + 10FeSO4 + 8H2O → K2SO4 + 2MnSO4 + 5Fe2(SO4)3 + 8H2O
If 15.8g of potassium permanganate (VII) was used in the reaction, calculate the mass of iron (II) sulphate used in the above reaction.
Give one word or phrase for the following:
The ratio of the mass of a certain volume of gas to the mass of an equal volume of hydrogen under the same conditions of temperature and pressure.
Give two tests of the following:
Oxygen
Which of the following contains maximum number of molecules?
The vapour density of CH3OH is ______. (At. Wt. C = 12, H = 1, O = 16)
