Advertisements
Advertisements
प्रश्न
Calculate the ‘spin only’ magnetic moment of \[\ce{M^{2+}_{ (aq)}}\] ion (Z = 27).
उत्तर
The electronic configuration of the M atom (Z = 27) is [Ar] 3d7 4s2.
∴ Electronic configuration of M2+ = [Ar] 3d7
i.e.,
| ↑↓ | ↑↓ | ↑ | ↑ | ↑ |
So it has three unpaired electrons.
∴ 'Spin-only' magnetic moment is calculated by using formula,
(µ) = `sqrt("n"("n" + 2))` B.M.
= `sqrt(3(3 + 2))` B.M.
= `sqrt15` B.M.
= 3.87 B.M.
APPEARS IN
संबंधित प्रश्न
Account for the following:
Cu+2 salts are coloured, while Zn2+ salts are white.
What are the transition elements? Write two characteristics of the transition elements.
How would you account for the following:
Cobalt (II) is stable in aqueous solutions, but in the presence of complexing reagents, it is easily oxidised.
Which metal in the first series of transition metals exhibits +1 oxidation state most frequently and why?
What can be inferred from the magnetic moment value of the following complex species?
| Example | Magnetic Moment (BM) |
| K2[MnCl4] | 5.9 |
Following are the transition metal ions of 3d series:
Ti4+, V2+, Mn3+, Cr3+
(Atomic numbers: Ti = 22, V = 23, Mn = 25, Cr = 24)
Answer the following:
1) Which ion is most stable in an aqueous solution and why?
2) Which ion is a strong oxidising agent and why?
3) Which ion is colourless and why?
Explain why transition metals and their compounds act as a catalyst.
Electronic configuration of a transition element X in +3 oxidation state is [Ar]3d5. What is its atomic number?
Transition elements form binary compounds with halogens. Which of the following elements will form \[\ce{MF3}\] type compounds?
(i) \[\ce{Cr}\]
(ii) \[\ce{Co}\]
(iii) \[\ce{Cu}\]
(iv) \[\ce{Ni}\]
Which of the following will not act as oxidising agents?
(i) \[\ce{CrO3}\]
(ii) \[\ce{MoO3}\]
(iii) \[\ce{WO3}\]
(iv) \[\ce{CrO^{2-}4}\]
Why EΘ values for Mn, Ni and Zn are more negative than expected?
Answer the following question:
Which element of the first transition series has highest second ionisation enthalpy?
Which of the following is non-metallic?
Mercury is the only metal liquid at room temperature due to its:-
Which of the following ions will exhibit colour in aqueous solution?
Account for the following:
Eu2+ with electronic configuration [Xe]4f76s2 is a strong reducing agent.
The trend of which property is represented by the following graph?
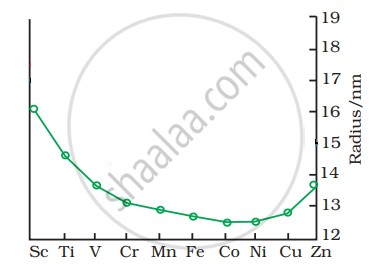
In order to protect iron from corrosion, which one will you prefer as a sacrificial electrode, Ni or Zn? Why? (Given standard electrode potentials of Ni, Fe and Zn are -0.25 V, -0.44 V and -0.76 V respectively.)
Decide which of the following atomic numbers are the atomic numbers of the inner transition elements:
29, 59, 74, 95, 102, 104
Compare the general characteristics of the first series of the transition metals with those of the second and third series metals in the respective vertical columns. Give special emphasis on the following point:
Oxidation states
