Advertisements
Advertisements
प्रश्न
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCl -> \underline{\phantom{..........}}}\].
उत्तर
\[\ce{NH4OH + HCl (dil{.}) -> \underline{\ce{NH4Cl + H2O}}}\].
APPEARS IN
संबंधित प्रश्न
Name a black metallic oxide which reacts with hydrochloric acid to give a coloured solution.
Complete the following reaction and balance them.
Zn+ 2HCl ⟶
Calcium oxide and phosphorous pentoxide are very good drying agents but they are not used to dry HCI gas. Give reasons for each.
Name the gas evolved when dilute hydrochloric acid is added to: Calcium carbonate
Give a balanced equation when dilute hydrochloric acid is added to : Calcium carbonate
Write an equation for the reaction of hydrochloric acid on:
Manganese (IV) oxide
Complete and balance the following reaction, state whether dilute or cone. acid is used.
\[\ce{NA4OH + HCl -> }\]
Complete and balance the following reaction, state whether dilute or cone. acid is used.
\[\ce{NH4OH + HCl ->}\]
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
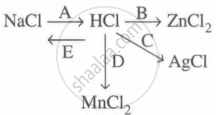
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.

