Advertisements
Advertisements
प्रश्न
Complete the following reaction and justify that it is a disproportionation reaction:
\[\ce{3MnO^{2-}4 + 4H^+ -> \underline{}\underline{}\underline{}\underline{} + \underline{}\underline{}\underline{}\underline{} + 2H2O}\]
उत्तर
\[\ce{3MnO^{2-}4 + 4H^+ -> 2MnO^-4 + MnO2 + 2H2O}\]
This is a disproportionation reaction since the same element [Mn] is oxidised and reduced in this process.
APPEARS IN
संबंधित प्रश्न
Which metal in the first transition series (3d series) exhibits + 1 oxidation state most frequently and why?
Calculate the ‘spin only’ magnetic moment of \[\ce{M^{2+}_{ (aq)}}\] ion (Z = 27).
An antifriction alloy made up of antimony with tin and copper, which is extensively used in machine bearings is called _______.
(A) Duralumin
(B) Babbitt metal
(C) Spiegeleisen
(D) Amalgam
Electronic configuration of a transition element X in +3 oxidation state is [Ar]3d5. What is its atomic number?
Transition elements show high melting points. Why?
Although \[\ce{Cr^3+}\] and \[\ce{Co^2+}\] ions have same number of unpaired electrons but the magnetic moment of \[\ce{Cr^3+}\] is 3.87 B.M. and that of \[\ce{Co^2+}\] is 4.87 B.M. Why?
EΘ of Cu is + 0.34V while that of Zn is – 0.76V. Explain.
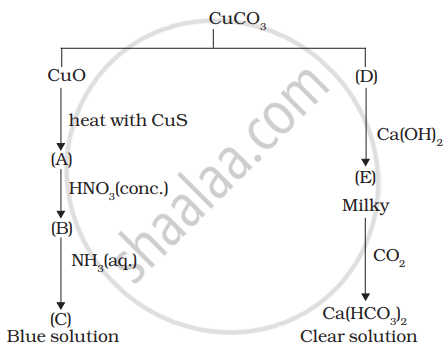
Identify A to E and also explain the reactions involved.
Explain the use of different transition metals as catalysts.
Give a reason for the following:
Zinc, cadmium and mercury are considered as d-block elements but not regarded as transition elements.
