Advertisements
Advertisements
प्रश्न
Consider Figure for photoemission.
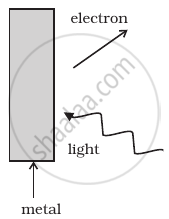
How would you reconcile with momentum conservation? Note light (photons) have momentum in a different direction than the emitted electrons.
उत्तर
The momentum is transferred to the metal. At the microscopic level, atoms absorb the photon and its momentum is transferred mainly to the nucleus and electrons. The excited electron is emitted. Conservation of momentum needs to be accounted for the momentum transferred to the nucleus and electrons.
APPEARS IN
संबंधित प्रश्न
Find the maximum frequency of X-rays produced by 30 kV electrons.
How does one explain the emission of electrons from a photosensitive surface with the help of Einstein's photoelectric equation?
Would you prefer a material with a high work-function or a low work-function to be used as a cathode in a diode?
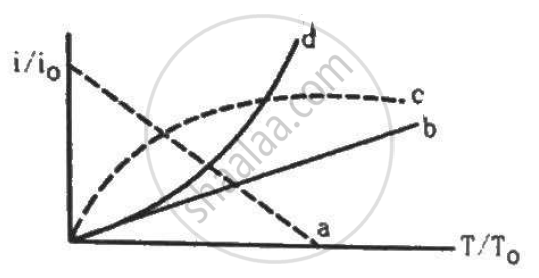
Let i0 be the thermionic current from a metal surface when the absolute temperature of the surface is T0. The temperature is slowly increased and the thermionic current is measured as a function of temperature. Which of the following plots may represent the variation in (i/i0) against (T/T0)?
The work function of aluminum is 4⋅2 eV. If two photons each of energy 2⋅5 eV are incident on its surface, will the emission of electrons take place? Justify your answer.
If a light of wavelength 330 nm is incident on a metal with work function 3.55 eV, the electrons are emitted. Then the wavelength of the wave associated with the emitted electron is (Take h = 6.6 × 10–34 Js)
Emission of electrons by the absorption of heat energy is called ____________ emission.
A 150 W lamp emits light of the mean wavelength of 5500 Å. If the efficiency is 12%, find out the number of photons emitted by the lamp in one second.
Name the factors on which photoelectric emission from a surface depends.
The work function of a metal is 2.31 eV. Photoelectric emission occurs when the light of frequency 6.4 × 1014 Hz is incident on the metal surface. Calculate
- the energy of the incident radiation,
- the maximum kinetic energy of the emitted electron and
- the stopping potential of the surface.
