Advertisements
Advertisements
प्रश्न
Consider Figure for photoemission.
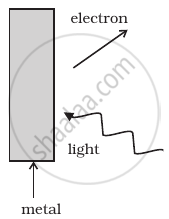
How would you reconcile with momentum conservation? Note light (photons) have momentum in a different direction than the emitted electrons.
उत्तर
The momentum is transferred to the metal. At the microscopic level, atoms absorb the photon and its momentum is transferred mainly to the nucleus and electrons. The excited electron is emitted. Conservation of momentum needs to be accounted for the momentum transferred to the nucleus and electrons.
APPEARS IN
संबंधित प्रश्न
The work function of caesium metal is 2.14 eV. When light of frequency 6 × 1014 Hz is incident on the metal surface, photoemission of electrons occurs. What is the
(a) maximum kinetic energy of the emitted electrons,
(b) Stopping potential, and
(c) maximum speed of the emitted photoelectrons?
An isolated metal sphere is heated to a high temperature. Will it become positively charged due to thermionic emission?
A diode value is connected to a battery and a load resistance. The filament is heated, so that a constant current is obtained in the circuit. As the cathode continuously emits electrons, does it become more and more positively charged?
Why does thermionic emission not take place in non-conductors?
The constant A in the Richardson−Dushman equation for tungsten is 60 × 104 A m−2K−2. The work function of tungsten is 4.5 eV. A tungsten cathode with a surface area 2.0 × 10−5 m2 is heated by a 24 W electric heater. In steady state, the heat radiated by the heater and the cathode equals the energy input by the heater and the temperature becomes constant. Assuming that the cathode radiates like a blackbody, calculate the saturation current due to thermions. Take Stefan's Constant = 6 × 10−8 W m−2 K−1. Assume that the thermions take only a small fraction of the heat supplied.
Define the term "Threshold frequency", in the context of photoelectric emission.
Answer the following question.
Define the term "Threshold frequency", in the context of photoelectric emission.
The wave associated with a moving particle of mass 3 × 10–6 g has the same wavelength as an electron moving with a velocity 6 × 106 ms–1. The velocity of the particle is
Why do metals have a large number of free electrons?
Name the factors on which photoelectric emission from a surface depends.
