Advertisements
Advertisements
प्रश्न
Why do metals have a large number of free electrons?
उत्तर
In metals, the electrons in the outermost shells are loosely bound to the nucleus. Even at room temperature, there are a large number of free electrons which are moving inside the metal in a random manner.
APPEARS IN
संबंधित प्रश्न
Find the maximum frequency of X-rays produced by 30 kV electrons.
Quarks inside protons and neutrons are thought to carry fractional charges [(+2/3)e; (–1/3)e]. Why do they not show up in Millikan’s oil-drop experiment?
The work function of the following metals is given : Na 2.75 ev, K = 2.3 eV, Mo = 4.17 eV and Ni = 5.15 eV. Which of these metals will not cause photoelectric emission for radiation of wavelength 3300 Å from a laser source placed 1 m away from these metals? What happens if the laser source is brought nearer and placed 50 cm away?
A diode value is connected to a battery and a load resistance. The filament is heated, so that a constant current is obtained in the circuit. As the cathode continuously emits electrons, does it become more and more positively charged?
Why does thermionic emission not take place in non-conductors?
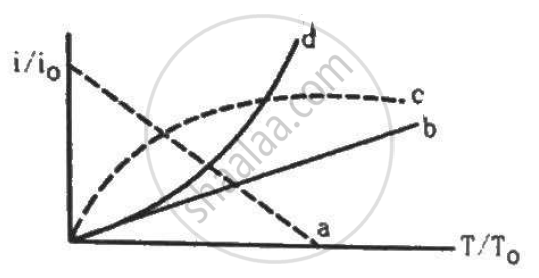
Let i0 be the thermionic current from a metal surface when the absolute temperature of the surface is T0. The temperature is slowly increased and the thermionic current is measured as a function of temperature. Which of the following plots may represent the variation in (i/i0) against (T/T0)?
If a light of wavelength 330 nm is incident on a metal with work function 3.55 eV, the electrons are emitted. Then the wavelength of the wave associated with the emitted electron is (Take h = 6.6 × 10–34 Js)
What do you mean by electron emission? Explain briefly various methods of electron emission.
Consider Figure for photoemission.
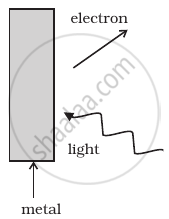
How would you reconcile with momentum conservation? Note light (photons) have momentum in a different direction than the emitted electrons.
Give an example each of a metal from which photoelectric emission takes place when irradiated by
- UV light
- visible light.
