Advertisements
Advertisements
प्रश्न
Give an example each of a metal from which photoelectric emission takes place when irradiated by
- UV light
- visible light.
उत्तर
- Photoelectric emission takes place when Zn is irradiated with UV radiation.
- Photoelectric emission takes place when Na is irradiated with visible light.
APPEARS IN
संबंधित प्रश्न
Find the maximum frequency of X-rays produced by 30 kV electrons.
How does one explain the emission of electrons from a photosensitive surface with the help of Einstein's photoelectric equation?
A diode value is connected to a battery and a load resistance. The filament is heated, so that a constant current is obtained in the circuit. As the cathode continuously emits electrons, does it become more and more positively charged?
Why does thermionic emission not take place in non-conductors?
Let i0 be the thermionic current from a metal surface when the absolute temperature of the surface is T0. The temperature is slowly increased and the thermionic current is measured as a function of temperature. Which of the following plots may represent the variation in (i/i0) against (T/T0)?
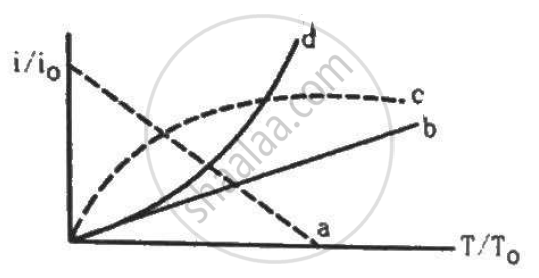
The anode of a thermionic diode is connected to the negative terminal of a battery and the cathode to its positive terminal.
The constant A in the Richardson−Dushman equation for tungsten is 60 × 104 A m−2K−2. The work function of tungsten is 4.5 eV. A tungsten cathode with a surface area 2.0 × 10−5 m2 is heated by a 24 W electric heater. In steady state, the heat radiated by the heater and the cathode equals the energy input by the heater and the temperature becomes constant. Assuming that the cathode radiates like a blackbody, calculate the saturation current due to thermions. Take Stefan's Constant = 6 × 10−8 W m−2 K−1. Assume that the thermions take only a small fraction of the heat supplied.
Answer the following question.
Define the term "Threshold frequency", in the context of photoelectric emission.
The wavelength λe of an electron and λp of a photon of same energy E are related by
If a light of wavelength 330 nm is incident on a metal with work function 3.55 eV, the electrons are emitted. Then the wavelength of the wave associated with the emitted electron is (Take h = 6.6 × 10–34 Js)
