Advertisements
Advertisements
प्रश्न
The wavelength λe of an electron and λp of a photon of same energy E are related by
पर्याय
`λ_"p" ∝ λ_"e"`
`λ_"p" ∝ sqrt(λ_"e")`
`λ_"p" ∝ 1/sqrt(λ_"e")`
`λ_"p" ∝ λ_"e"^2`
उत्तर
`λ_"p" ∝ λ_"e"^2`
APPEARS IN
संबंधित प्रश्न
Quarks inside protons and neutrons are thought to carry fractional charges [(+2/3)e; (–1/3)e]. Why do they not show up in Millikan’s oil-drop experiment?
How does one explain the emission of electrons from a photosensitive surface with the help of Einstein's photoelectric equation?
The work function of the following metals is given : Na 2.75 ev, K = 2.3 eV, Mo = 4.17 eV and Ni = 5.15 eV. Which of these metals will not cause photoelectric emission for radiation of wavelength 3300 Å from a laser source placed 1 m away from these metals? What happens if the laser source is brought nearer and placed 50 cm away?
Would you prefer a material with a high melting point or a low melting point to be used as a cathode in a diode?
A diode value is connected to a battery and a load resistance. The filament is heated, so that a constant current is obtained in the circuit. As the cathode continuously emits electrons, does it become more and more positively charged?
Let i0 be the thermionic current from a metal surface when the absolute temperature of the surface is T0. The temperature is slowly increased and the thermionic current is measured as a function of temperature. Which of the following plots may represent the variation in (i/i0) against (T/T0)?
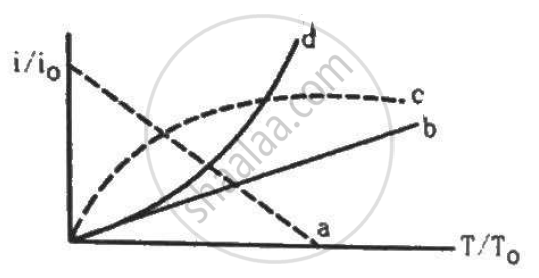
Answer the following question.
Define the term "Threshold frequency", in the context of photoelectric emission.
If a light of wavelength 330 nm is incident on a metal with work function 3.55 eV, the electrons are emitted. Then the wavelength of the wave associated with the emitted electron is (Take h = 6.6 × 10–34 Js)
Why do metals have a large number of free electrons?
Name the factors on which photoelectric emission from a surface depends.
