Advertisements
Advertisements
प्रश्न
Consider the following organic compounds:
HCHO, C2H5OH, C2H6, CH3COOH, C2H5CI
Choose two compounds which can react in the presence of conc. H2SO4 to form an ester. Give the name and formula of the ester formed.
उत्तर
C2H5OH (ethanol) and CH3COOH (ethanoic acid) are the compounds which can react with each other, in the presence of concentrated sulphuric acid, to form a sweet smelling ester called ethylethanoate. Its formula is CH3 -COO-CH2-CH3.
The chemical equation for the above reaction is as follows
`C_2 H_5 OH + CH_3 COOH` 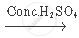 `CH_3 COOC_2 H_5 + H_2 O`
`CH_3 COOC_2 H_5 + H_2 O`
APPEARS IN
संबंधित प्रश्न
Consider the following comments about saponification reactions:
I. Heat is evolved in these reactions.
II. For quick precipitation of soap, sodium chloride is added to the reaction mixtures.
III. Saponification reactions are a special kind of neutralisation reactions.
IV. Soaps are basic salts of long-chain fatty acids.
The correct comments are
(a) I, II and III
(b) II, III and IV
(c) I, II and IV
(d) Only I and IV
What are esters ?
Give the common names and IUPAC names of the following compounds of CH3COOH.
Give the common name and IUPAC name of C2H5OH.
Name one chemical compound which can be used to distinguish between ethanol and ethanoic acid.
What will you observe when dilute ethanoic acid and dilute hydrochloric acid are put on universal indicator paper, one by one? What does it show?
What happens when ethanoic acid reacts with sodium carbonate? Write chemical equation of the reaction involved.
How Will You Carry Out the Following Conversions?
Ehane to acitic acid
Observe the figure and write the answers to the following questions.

- Write the name of the reaction shown in the following figure.
- Write the above chemical reaction in the form of a balanced equation.
- Write the name of the product produced in the above reaction, write a use.
- Write the name of the catalyst used in the above reaction.
Anita added a drop each of diluted acetic acid and diluted hydrochloric acid on pH paper and compared the colors. Which of the following is the correct conclusion?
