Advertisements
Advertisements
प्रश्न
Consider the following organic compounds:
HCHO, C2H5OH, C2H6, CH3COOH, C2H5CI
Choose two compounds which can react in the presence of conc. H2SO4 to form an ester. Give the name and formula of the ester formed.
उत्तर
C2H5OH (ethanol) and CH3COOH (ethanoic acid) are the compounds which can react with each other, in the presence of concentrated sulphuric acid, to form a sweet smelling ester called ethylethanoate. Its formula is CH3 -COO-CH2-CH3.
The chemical equation for the above reaction is as follows
`C_2 H_5 OH + CH_3 COOH` 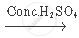 `CH_3 COOC_2 H_5 + H_2 O`
`CH_3 COOC_2 H_5 + H_2 O`
APPEARS IN
संबंधित प्रश्न
Complete the following chemical equations: C2H5OH+O2→
What is the common name of methanoic acid.
Draw the structure of butanoic acid.
What happens when ethanoic acid reacts with sodium carbonate? Write chemical equation of the reaction involved.
What is glacial acetic acid ?
Acetic acid is a typical acid. Write one equation in case of its reactions with a metal?
Explain the following term with example.
Reduction
Why is pure acetic acid also known as glacial acetic acid?
Ethanoic acid _________.
Ethanoic acid turns ______ litmus to ______
