Advertisements
Advertisements
प्रश्न
Consider the section of the periodic table given below :
| Group numbers |
IA | IIA | IIIA | IVA | VA | VIA | VIIA | O |
| 1 | 2 | 13 | 14 | 15 | 16 | 17 | 18 | |
| Li | D | O | J | Ne | ||||
| A | Mg | E | Si | H | K | |||
| B | C | F | G | L |
With reference to the table, answer the following :
(i) Which is the most electro negative ?
(ii) How many valence electrons are present in G ?
(iii) Write the formula of the compound between B and H ?
(iv) In the compound between F and J, what type of bond will be formed ?
(v) Draw the electron dot structure for the compound formed between C and K.
उत्तर
(i) K is the most electro negative.
(ii) 5 valence electrons are present in G.
(iii) B2H
(iv) Ionic
(v)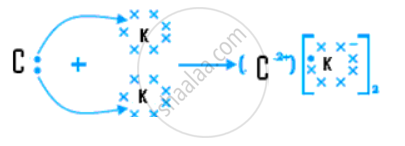
APPEARS IN
संबंधित प्रश्न
Rewrite the following statement after correction, if necessary:
The metallic character of elements in a period increases gradually on moving from left to right.
Complete the following sentences
The similarities in the properties of a group of elements are because they have the same …………………
Name the element in period 3 which does not form oxide.
How many groups and periods are there in the modern periodic table?

In the above table, H does not represent hydrogen.Some elements are given in their own symbol and position in the periodic table while others are shown with a letter. With refrence to the table answer the following questions.
1. Identify the most electronegative element.
2. Identify the most reactive element of Group I.
3. Identify the element from Period 3 with least atomic size.
4. How many valence electrons are present in Q?
5. Which element from group 2 would have the least ionisation energy?
6. Identify the noble gas of the fourth period.
7. In the compound between A and H, what type of bond would be formed and give its molecular formula.
Arrange the following as per instruction given in the bracket
Na, K, Cl, S, Si (increasing ionisation potential)
Arrange the following as per instruction given in the bracket.
Cl, F, Br, I (increasing electron affinity)
Arrange the following in order of increasing radii:
Mg2+, Mg, Mg+
State the nature of compounds formed when group 17 elements combine with (i) metals (ii) non-metals.
Write the electronic configuration of two elements A and B whose atomic numbers are 20 and 17 respectively. Write the molecular formula of the compound formed when element A reacts with element B. State whether this compound is acidic, basic or neutral. Give a reason to justify your answer.
