Advertisements
Advertisements
Question
Consider the section of the periodic table given below :
| Group numbers |
IA | IIA | IIIA | IVA | VA | VIA | VIIA | O |
| 1 | 2 | 13 | 14 | 15 | 16 | 17 | 18 | |
| Li | D | O | J | Ne | ||||
| A | Mg | E | Si | H | K | |||
| B | C | F | G | L |
With reference to the table, answer the following :
(i) Which is the most electro negative ?
(ii) How many valence electrons are present in G ?
(iii) Write the formula of the compound between B and H ?
(iv) In the compound between F and J, what type of bond will be formed ?
(v) Draw the electron dot structure for the compound formed between C and K.
Solution
(i) K is the most electro negative.
(ii) 5 valence electrons are present in G.
(iii) B2H
(iv) Ionic
(v)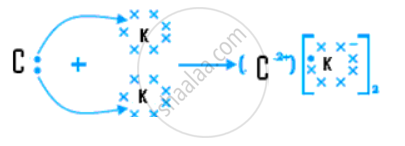
APPEARS IN
RELATED QUESTIONS
The atomic numbers of three elements A, B and C are 12, 18 and 20 respectively. State, giving reason, which two elements will show similar properties.
The elements Be, Mg and Ca each having two electrons in their outermost shells are in periods 2, 3, and 4 respectively of the modern periodic table. Answer the following questions, giving justification in each case:-
(i) Write the group to which these elements belong.
(ii) Name the least reactive element.
(iii) Name the element having largest atomic radius.
How does the size of atoms (atomic size) generally vary in going from left to right in a period of the periodic table? Why does it vary this way?
What do you understand by periodicity?
State the valency of the elements of periods 3 and write the formula of their oxides.
K, Pb, Ca, Zn (In the increasing order of the reactivity)
Name the elements in period 1.
Choose the correct answer from the choice given:
An element A belonging to Period 3 and Group II will have
With reference to the variation of properties in the Periodic table, which of the following is generally true?
Based on the group valency of element write the molecular formula of the following compound giving justification:
Compound formed when an element A of group 2 combines with an element B of group seventeen.
