Advertisements
Advertisements
प्रश्न
Consider the section of the periodic table given below :
| Group numbers |
IA | IIA | IIIA | IVA | VA | VIA | VIIA | O |
| 1 | 2 | 13 | 14 | 15 | 16 | 17 | 18 | |
| Li | D | O | J | Ne | ||||
| A | Mg | E | Si | H | K | |||
| B | C | F | G | L |
With reference to the table, answer the following :
(i) Which is the most electro negative ?
(ii) How many valence electrons are present in G ?
(iii) Write the formula of the compound between B and H ?
(iv) In the compound between F and J, what type of bond will be formed ?
(v) Draw the electron dot structure for the compound formed between C and K.
उत्तर
(i) K is the most electro negative.
(ii) 5 valence electrons are present in G.
(iii) B2H
(iv) Ionic
(v)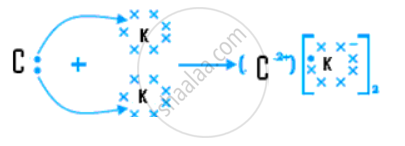
APPEARS IN
संबंधित प्रश्न
The atomic masses of three elements A, B and C having similar chemical properties are 7, 23 and 39, respectively.
- Calculate the average atomic mass of elements A and C.
- Compare the average atomic mass with atomic mass of B.
- What could the elements A, B and C be?
An element 'M' with electronic configuration (2, 8, 2) combines separately with (NO3)–, (SO4)2– and (PO4)3– radicals. Write the formula of the three compounds so formed. To which group and period of the Modern Periodic Table does the element 'M' belong? Will 'M' form covalent or ionic compounds? Give reason to justify your answer.
Name any two elements of group one and write their electronic configurations. What similarity do you observe in their electronic configurations? Write the formula of oxide of any of the aforesaid elements.
The atomic number of an element is 20. In modern periodic table, this element is placed:
(a) 2nd period
(b) 4th period
(c) 3rd period
(d) 1st period
How do the following change on moving from left to right in a period of the periodic table?
Give examples in support of your answer.
Chemical reactivity of elements ?
Choose the most appropriate answer from the following list of oxides which fit the description.
An amphoteric oxide: Al2O3 (shows both acidic and basic properties)
Arrange the following as per instruction given in the bracket.
Li, K, Na, H ( decreasing order of their potential ionisation )
Name the elements in Period 1.
Name or state following with reference to the element of the first three periods of the periodic table.
A non-metal in period 2 having electronic configuration 2, 6.
Electron Affinity is maximum in ______.
