Advertisements
Advertisements
प्रश्न
Consider the change in the oxidation state of Bromine corresponding to different emf values as shown in the diagram below:
\[\ce{BrO^-_4 ->[1.82 V] BrO^-_3 ->[1.5 V] HBrO ->[1.595 V] Br2 ->[1.0652 V] Br^-}\]
Then the species undergoing disproportionation is
विकल्प
Br2
\[\ce{BrO^-_4}\]
\[\ce{BrO^-_3}\]
HBrO
उत्तर
HBrO
Explanation:
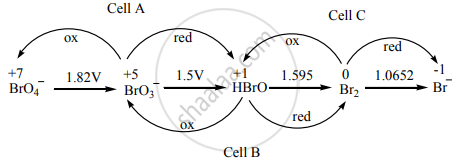
(Ecell)A = – 1.82 + 1.5 = – 0.32 V
(Ecell)B = – 1.5 + 1.595 = + 0.095 V
(Ecell)C = 1.595 + 1.0652 = – 0.529 V
∴ The species undergoing disproportionation is HBrO.
APPEARS IN
संबंधित प्रश्न
Write cathode and anode reaction in a fuel cell.
If one mole electrons is passed through the solutions of CrCl3, AgNO3 and NiSO4, in what ratio Cr, Ag and Ni will be deposited at the electrodes?
Describe the construction of Daniel cell. Write the cell reaction.
A copper electrode is dipped in 0.1 M copper sulphate solution at 25°C. Calculate the electrode potential of copper.
[Given: \[\ce{E^0_{{Cu^{2+}|Cu}}}\] = 0.34 V]
For the cell \[\ce{Mg_{(s)}|Mg^{2+}_{( aq)}||Ag^+_{( aq)}|Ag_{(s)}}\], calculate the equilibrium constant at 25°C and maximum work that can be obtained during operation of cell.
Given: \[\ce{E^0_{{Mg^{2+}|Mg}}}\] = −2.37 V and \[\ce{E^0_{{Ag^{+}|Ag}}}\] = 0.80 V
The electrochemical cell stops working after some time because
Which of the following is incorrect?
The number of moles of electrons passed when the current of 2 A is passed through a solution of electrolyte for 20 minutes is ______.
The cell constant of a conductivity cell is 0.146 cm-1. What is the conductivity of 0.01 M solution of an electrolyte at 298 K, if the resistance of the cell is 1000 ohm?
State the term for the following:
Two metal plates or wires through which the current enters and leaves the electrolytic cell.
