Advertisements
Advertisements
प्रश्न
Consider the change in the oxidation state of Bromine corresponding to different emf values as shown in the diagram below:
\[\ce{BrO^-_4 ->[1.82 V] BrO^-_3 ->[1.5 V] HBrO ->[1.595 V] Br2 ->[1.0652 V] Br^-}\]
Then the species undergoing disproportionation is
पर्याय
Br2
\[\ce{BrO^-_4}\]
\[\ce{BrO^-_3}\]
HBrO
उत्तर
HBrO
Explanation:
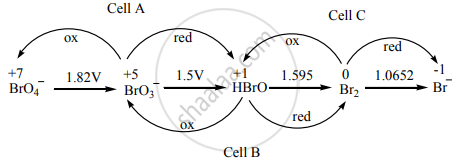
(Ecell)A = – 1.82 + 1.5 = – 0.32 V
(Ecell)B = – 1.5 + 1.595 = + 0.095 V
(Ecell)C = 1.595 + 1.0652 = – 0.529 V
∴ The species undergoing disproportionation is HBrO.
APPEARS IN
संबंधित प्रश्न
Write cathode and anode reaction in a fuel cell.
Why cannot we store AgNO3 solution in copper vessel?
If one mole electrons is passed through the solutions of CrCl3, AgNO3 and NiSO4, in what ratio Cr, Ag and Ni will be deposited at the electrodes?
How many faradays of electricity are required for the following reaction to occur
\[\ce{MnO^-_4 -> Mn^2+}\]
A gas X at 1 atm is bubbled through a solution containing a mixture of 1MY− and 1MZ− at 25°C. If the reduction potential of Z > Y > X, then ____________.
Why is anode in galvanic cell considered to be negative and cathode positive electrode?
Two metals M1 and M2 have reduction potential values of −xV and +yV respectively. Which will liberate H2 and H2SO4.
What is electrode potential?
Assertion: ECell should have a positive value for the cell to function.
Reason: `"E"_("cathode") < "E"_("anode")`
Explain the types of electrochemical cells.
