Advertisements
Advertisements
प्रश्न
Consider two elements 'A' (Atomic number 17) and 'B' (Atomic number 19) :
(i) Write the positions of these elements in the modern periodic table giving justification.
(ii) Write the formula of the compound formed when 'A' combines with 'B.'
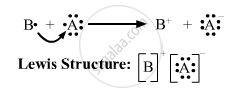
(iii) Draw the electron dot structure of the compound and state the nature of the bond formed between the two elements.
उत्तर
(i) Position of the elements in the periodic table:
| Element | Period | Group |
| A | 3 | 17 |
| B | 4 | 1 |
(ii)
Atomic number of A = 17
Electronic configuration A = 2,8,7
Number of valence electrons of A = 7
Valency of A = 8 - 7 = 1
Atomic number of B = 19
Electronic configuration B = 2,8,8,1
Number of valence electrons of B = 1
Valency of A = 1
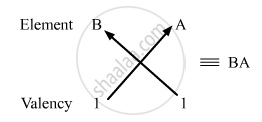
So, the formula of the compound formed when elements A and B combine is BA.
(iii)
APPEARS IN
संबंधित प्रश्न
Use the letters only written in the Periodic Table given below to answer the questions that Follow :

1) State the number of valence electrons in atom J.
2) Which element shown forms ions with a single negative charge?
3)Which metallic element is more reactive than R?
4) Which element has its electrons arranged in four shells?
In which part of a group would you separately expect the elements to have the largest atomic size?
Why does the size of the atoms progressively become smaller when we move from sodium (Na) to chlorine (Cl) in the third period of the periodic table?
Name two properties of elements whose magnitudes change when going from top to bottom in a group of the periodic table. In what manner do they change?
Which of the following set of elements is written correctly in the order of their increasing metallic character?
(a) Mg, Al, Si
(b) C, O, N
(c) Na, Li, K
(d) Be, Mg, Ca
An element X belong to 4th period and 17th group, state.
no of valence electrons in it
Calculate the valency of element X whose atomic number is 9.
Give the number of the group and the period of elements having three shells with three electrons in valence shell.
Answer the following question:
State the four blocks of the modern periodic table based on the electronic configuration of elements.
Name the elements in Period 1.
