Advertisements
Advertisements
Question
Consider two elements 'A' (Atomic number 17) and 'B' (Atomic number 19) :
(i) Write the positions of these elements in the modern periodic table giving justification.
(ii) Write the formula of the compound formed when 'A' combines with 'B.'
(iii) Draw the electron dot structure of the compound and state the nature of the bond formed between the two elements.
Solution
(i) Position of the elements in the periodic table:
| Element | Period | Group |
| A | 3 | 17 |
| B | 4 | 1 |
(ii)
Atomic number of A = 17
Electronic configuration A = 2,8,7
Number of valence electrons of A = 7
Valency of A = 8 - 7 = 1
Atomic number of B = 19
Electronic configuration B = 2,8,8,1
Number of valence electrons of B = 1
Valency of A = 1
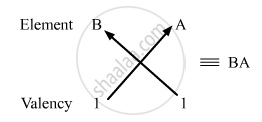
So, the formula of the compound formed when elements A and B combine is BA.
(iii)
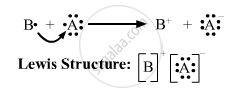
APPEARS IN
RELATED QUESTIONS
The metal of Group 2 from top to bottom arc Be, Mg, Ca, Sr, and Ba.
1) Which one of these elements will form ions most readily and why?
2) State the common feature in the electronic configuration of all these elements.
How does the valency of elements change of moving from left to right in the third period of the periodic table?
Name the element which is in seventeenth group and second period.
Given alongside is a part of the periodic table. As we move vertically downward from Li to Fr:
| Li | Be |
| Na | |
| K | |
| Rb | |
| Cs | |
| Fr | Ra |
What happens to their metallic character?
Four elements P, Q, R and S belong to the third period of the Modern Periodic Table and have respectively 1, 3, 5 and 7 electrons in their outermost shells. Write the electronic configurations of Q and R and determine their valences. Write the molecular formula of the compound formed when P and S combine.
Explain, the statement 'In each period, the atomic radii gradually decrease with increase in atomic number'. Give one example to justify your answer.
The elements of one short period of the periodic table are given below in order from left to right:
| Li | Be | B | C | O | F | Ne |
To which period do these elements belong?
Supply the missing word from those in bracket:
If an element has one electron in its outermost energy level [shell] then it is likely to be _______.
Name or state following with reference to the elements of the first three periods of the periodic table.
A metalloid in period 2 and in period 3.
Name or state following with reference to the elements of the first three periods of the periodic table.
The number of electron shells in elements of period 1, period 2, and period 3.
