Advertisements
Advertisements
Question
Four elements P, Q, R and S belong to the third period of the Modern Periodic Table and have respectively 1, 3, 5 and 7 electrons in their outermost shells. Write the electronic configurations of Q and R and determine their valences. Write the molecular formula of the compound formed when P and S combine.
Solution
| Elements | Valence Electrons |
| P | 1 |
| Q | 3 |
| R | 5 |
| S | 7 |
Electronic configuration of Q = 2, 8, 3
If the number of electrons in the valence shell is less than 4, then the same number of electrons will represent the valency of that element.
Valency of Q = 3
Electronic configuration of R = 2, 8, 5
If the number of valence electrons is either equal to or greater than 4, then that number is subtracted from 8 to obtain the correct valency of that element.
Valency of R = 8 − 5 = 3
Molecular formula of the compound formed when P and S combine:
Number of valence electrons in P = 1
Valency of P = 1
Number of valence electrons in S = 7
Valency of S = 8 − 7 = 1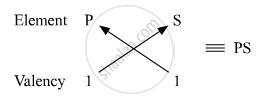
APPEARS IN
RELATED QUESTIONS
Write the number of vertical columns in the modern periodic table. What are these columns called?
Use the letters only written in the Periodic Table given below to answer the questions that Follow :

1) State the number of valence electrons in atom J.
2) Which element shown forms ions with a single negative charge?
3)Which metallic element is more reactive than R?
4) Which element has its electrons arranged in four shells?
How does the number of valence electrons vary on moving from left or right in the second period of the periodic table?
What is its atomic number?
K, Pb, Ca, Zn (In the increasing order of the reactivity)
The following table represents the first period of the modern periodic table. Study the table and answer the questions that follow:
(i) Write the formula of the sulphate of the element with atomic number 13.
(ii) What type of bonding will be present in the oxide of the element with atomic number 1?
(iii) Which feature of the atomic structure accounts for the similarities in the chemical properties of the elements in group VIIA of the periodic table?
(iv) Name the element which has the highest ionization potential.
(v) How many electrons are present in the valence shell of the element with atomic number 18?
(vi) What is the name given to the energy released, when an atom in its isolated gaseous state accepts an electron to form an anion?
(vii) What is the electronic configuration of the element in the third period which gains one electron to become an anion?
(viii) Fill in the blanks:
The atomic size ______ as we move from left to right across the period, because the ______ increases, but the ______ remains the same.
Choose the most appropriate answer from the following list of oxides which fit the description.
An oxide which dissolves in water forming an acid.
Atomic numbers of elements A, B, C, D, E, F are 8, 7, 11, 12, 13 and 9 respectively. State the type of ions they form.
An element A has 2 electrons in its fourth shell. State:
its valency
What is the common feature of the electronic configuration of the elements at the end of Period 2 and Period 3?
