Advertisements
Advertisements
प्रश्न
Four elements P, Q, R and S belong to the third period of the Modern Periodic Table and have respectively 1, 3, 5 and 7 electrons in their outermost shells. Write the electronic configurations of Q and R and determine their valences. Write the molecular formula of the compound formed when P and S combine.
उत्तर
| Elements | Valence Electrons |
| P | 1 |
| Q | 3 |
| R | 5 |
| S | 7 |
Electronic configuration of Q = 2, 8, 3
If the number of electrons in the valence shell is less than 4, then the same number of electrons will represent the valency of that element.
Valency of Q = 3
Electronic configuration of R = 2, 8, 5
If the number of valence electrons is either equal to or greater than 4, then that number is subtracted from 8 to obtain the correct valency of that element.
Valency of R = 8 − 5 = 3
Molecular formula of the compound formed when P and S combine:
Number of valence electrons in P = 1
Valency of P = 1
Number of valence electrons in S = 7
Valency of S = 8 − 7 = 1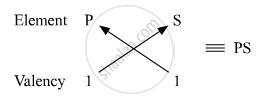
APPEARS IN
संबंधित प्रश्न
The atomic masses of three elements A, B and C having similar chemical properties are 7, 23 and 39, respectively.
- Calculate the average atomic mass of elements A and C.
- Compare the average atomic mass with atomic mass of B.
- What could the elements A, B and C be?
Consider two elements 'A' (Atomic number 17) and 'B' (Atomic number 19) :
(i) Write the positions of these elements in the modern periodic table giving justification.
(ii) Write the formula of the compound formed when 'A' combines with 'B.'
(iii) Draw the electron dot structure of the compound and state the nature of the bond formed between the two elements.
Which element has twice as many electrons in its second shell as in its first shell?
Fill in the blank in the following statement:
In going across a period (right to left) in periodic table, the atomic size of the atom ...............
How does the electropositive character of elements change on going down in a group of the periodic table?
State one reason for keeping fluorine and chlorine in the same of the periodic table.
Name two properties of elements whose magnitudes change when going from top to bottom in a group of the periodic table. In what manner do they change?
How do the following change on moving from left to right in a period of the periodic table?
Give examples in support of your answer.
atomic structure (electron arrangements) ?
An element 'X' (Atomic number – 20) burns in the presence of oxygen to form a basic oxide.
(a) Identify the element and write its electronic configuration.
(b) State its group number and period number in the Modern Periodic Table.
(c) Write a balanced chemical equation for the reaction when this oxide is dissolved in water.
Select the correct answer
Identify the metalloid
