Advertisements
Advertisements
प्रश्न
Four elements P, Q, R and S belong to the third period of the Modern Periodic Table and have respectively 1, 3, 5 and 7 electrons in their outermost shells. Write the electronic configurations of Q and R and determine their valences. Write the molecular formula of the compound formed when P and S combine.
उत्तर
| Elements | Valence Electrons |
| P | 1 |
| Q | 3 |
| R | 5 |
| S | 7 |
Electronic configuration of Q = 2, 8, 3
If the number of electrons in the valence shell is less than 4, then the same number of electrons will represent the valency of that element.
Valency of Q = 3
Electronic configuration of R = 2, 8, 5
If the number of valence electrons is either equal to or greater than 4, then that number is subtracted from 8 to obtain the correct valency of that element.
Valency of R = 8 − 5 = 3
Molecular formula of the compound formed when P and S combine:
Number of valence electrons in P = 1
Valency of P = 1
Number of valence electrons in S = 7
Valency of S = 8 − 7 = 1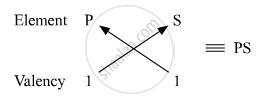
APPEARS IN
संबंधित प्रश्न
Match the atomic number 2, 4, 8, 15 and 19 with each of the following
1) A solid non metal belonging to the third period.
2) A metal of valency 1.
3) A gaseous element with valency 2.
4) An element belonging to Group 2
5) A rare gas.
An element Z has atomic number 16. Answer the following questions on Z:
1) State the period and group to which Z belongs
2) Is Z a metal or a non-metal?
3) State the formula between Z and hydrogen
4) What kind of a compound is this?
Why do elements in the same group show similar properties but the elements in different groups show different properties?
Name two elements you would expect to show chemical reactions similar to calcium. What is the basis of your choice?
Complete the following sentences.
The properties of the elements are a periodic function of their …………… (atomic number, mass number, reative atomic mass).
Complete the following sentences
The similarities in the properties of a group of elements are because they have the same …………………
Choose the most appropriate answer from the following list of oxides which fit the description.
An amphoteric oxide.
Name or state following with reference to the element of the first three periods of the periodic table.
The formula of the hydroxide of the element having electronic configuration 2, 8, 2.
Fill in the blanks from the words A to F given below’.
A: Decreases
B: Increases
C: Remains same
D: Increases by one
E: Electropositive
F: Electronegative
Across a period from left to right in the Modern Periodic Table.
No. of electron shells _________; No. of valence electrons __________: Electronegativity increases Character of elements changes from electropositive to ________.
Electron Affinity is maximum in ______.
