Advertisements
Advertisements
प्रश्न
Copy and complete the following table which refers to the industrial method for the preparation of ammonia and sulphuric acid:
| Name of the compoun | Name of the process | Catalytic equation (with the catalyst) |
| Ammonia | (i)__________ | (ii) __________ |
| Sulhuric acid | (iii) ___________ | (iv) __________ |
उत्तर
| Name of the compound | Name of the process | Catalytic equation (with the catalyst) |
| Ammonia | (i)Haber process | 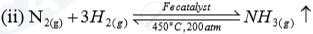 |
| Sulhuric acid | (iii)Contact process |  |
APPEARS IN
संबंधित प्रश्न
Identify the substance underlined:
The catalyst used to oxide ammonia.
Name the following
Liquid when added to metallic nitride that yield ammonia.
Write balanced chemical equations for the following:
An equation to illustrate the reducing nature of ammonia.
Describe all what you will observe and write chemical equation, when limited amount of ammonia gas is passed through the following aqueous solution:
FeCl3
(a) Name the catalyst used in 1(ii)
(b) In the reaction referred to in 1(ii), the catalyst glows red hot. Why?
(c) What is the name of the industrial process which starts with the reaction reffered to in 1 (ii)?
Industrially, ammonia is obtained by direct combination of nitrogen and hydrogen.
(i) Write balanced equation for the direct combination of nitrogen and hydrogen.
(ii) Which of the metals - iron, platinum, copper - catalyse this direct combination?
(iii) Is the formation of ammonia promoted by high pressure or low pressure?
Write balanced chemical equations for a reaction in which ammonia is oxidised by the following:
(i) A metal oxide
(ii) A gas which is not oxygen
Give balanced chemical equations for the following:
Reaction of ammonia with excess chlorine
How is ammonia dried and collected in the laboratory?
