Advertisements
Advertisements
Question
Copy and complete the following table which refers to the industrial method for the preparation of ammonia and sulphuric acid:
| Name of the compoun | Name of the process | Catalytic equation (with the catalyst) |
| Ammonia | (i)__________ | (ii) __________ |
| Sulhuric acid | (iii) ___________ | (iv) __________ |
Solution
| Name of the compound | Name of the process | Catalytic equation (with the catalyst) |
| Ammonia | (i)Haber process | 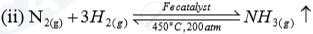 |
| Sulhuric acid | (iii)Contact process |  |
APPEARS IN
RELATED QUESTIONS
The equation 4NH3 + 5O2 → 4NO + 6 H2O, represents the catalytic oxidation of ammonia. If 100 cm3 of ammonia is used calculate the volume of oxygen required to oxidize the ammonia completely
Write balanced chemical equations for the following:
When the excess of ammonia is treated with chlorine.
State one relevant observation for the following
When ammonia gas is burnt in an atmosphere of excess oxygen.
Name the following
Salt known as ammoniac.
Give balanced chemical equations for the following:
Reaction of ammonia with sulphuric acid.
Name the substance used for drying ammonia gas? Why cannot substances such as conc.H2SO4, anhydrous calcium chloride and phosphorus pentoxide be used for drying ammonia gas?
Give balanced equation to prove that ammonia Is alkaline in nature
Describe all what you will observe and write chemical equation, when limited amount of ammonia gas is passed through the following aqueous solution:
Pb(NO3)2
(a) Name the catalyst used in 1(ii)
(b) In the reaction referred to in 1(ii), the catalyst glows red hot. Why?
(c) What is the name of the industrial process which starts with the reaction reffered to in 1 (ii)?
Write a balanced equation for the following conversion:
Nitrogen trichloride from ammonia.
