Advertisements
Advertisements
प्रश्न
Copy and complete the following table which refers to the industrial method for the preparation of ammonia and sulphuric acid:
| Name of the compoun | Name of the process | Catalytic equation (with the catalyst) |
| Ammonia | (i)__________ | (ii) __________ |
| Sulhuric acid | (iii) ___________ | (iv) __________ |
उत्तर
| Name of the compound | Name of the process | Catalytic equation (with the catalyst) |
| Ammonia | (i)Haber process | 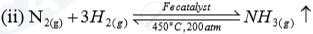 |
| Sulhuric acid | (iii)Contact process |  |
APPEARS IN
संबंधित प्रश्न
The acid which is prepared by catalytic oxidation of ammonia.
Name the gas obtained when ammonia burns in an atmosphere of oxygen without any catalyst.
Complete the following equation.
4 NH3 +5O2 →
What important process starts with the reaction in above? Name the catalyst used.
Give balanced chemical equations for each of the following:
Lab preparation of ammonia using an ammonium salt
Give balanced chemical equations for of the following :
Reaction of ammonia with excess chlorine.
Copy and complete the following equations :
(i) Mg3N2 + 6H2O →
(ii) 2NH3 + 3CuO →
(iii) 8NH3 + 3Cl2 →
(iv) 4NH3 + 5O2 →
(a) Name the catalyst used in 1(ii)
(b) In the reaction referred to in 1(ii), the catalyst glows red hot. Why?
(c) What is the name of the industrial process which starts with the reaction reffered to in 1 (ii)?
Write the equation for:
The preparation of ammonia from ammonium chloride and calcium hydroxide.
The catalyst used in the oxidation of ammonia is ______.
Ammonia burns in oxygen, as shown below.
\[\ce{4NH3 + 3O2 -> 2N2 + 6H2O}\]
If 240 cc of ammonia is burnt in 300 cc of oxygen, find out the composition of the resultant gaseous mixture at room temperature.
