Advertisements
Advertisements
प्रश्न
Distinguish between the following : Benzoic acid and methyl benzoate
उत्तर
Benzoic acid can react with sodium bicarbonate to give brisk effervescence due to the release of CO2, while methyl benzoate does not.

संबंधित प्रश्न
Although phenoxide ion has more number of resonating structures than carboxylate ion, carboxylic acid is a stronger acid than phenol. Give two reasons.
Why pKa of F-CH2-COOH is lower than that of Cl−CH2−COOH?
Which acid of the pair shown here would you expect to be stronger?
CH2FCO2H or CH2ClCO2H
Which acid of the pair shown here would you expect to be stronger?
CH2FCH2CH2CO2H or CH3CHFCH2CO2H
Which acid of the pair shown here would you expect to be stronger?
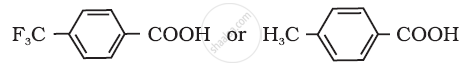
Arrange the following compounds in increasing order of their property as indicated:
CH3CH2CH(Br)COOH, CH3CH(Br)CH2COOH, (CH3)2CHCOOH, CH3CH2CH2COOH (acid strength)
Arrange the following compounds in increasing order of their property as indicated:
Benzoic acid, 4-Nitrobenzoic acid, 3, 4-Dinitrobenzoic acid, 4-Methoxybenzoic acid (acid strength)
Account for the following:
Carboxylic acid is a stronger acid than phenol.
Acidity of BF3 can be explained on the basis of which of the following concepts?
A mixture of benzaldehyde and formaldehyde on heating with 50% NaOH solution gives
