Advertisements
Advertisements
प्रश्न
Distinguish tetrahedral and octahedral voids.
अंतर स्पष्ट करें
उत्तर
| Sr. No. | Tetrahedral voids | Octahedral voids |
| 1. | A single triangular void in a crystal is surrounded by four (4) spheres and is called a tetrahedral void. | A double triangular void like c is surrounded by six (6) spheres and is called an octahedral void. |
| 2. | A sphere of second layer is above the void of the first layer, a tetrahedral void is formed. | The voids in the first layer are partially covered by the spheres of the layer. |
| 3. | This constitutes four spheres, three in the lower and one in the upper layer. When the centres of these four spheres are joined a tetrahedron is formed. | This constitutes six spheres, three in the lower layer and three in the upper layer. When the centers of these six spheres are joined an octahedron is formed. |
| 4. | The radius of the sphere which can be accommodated in an octahedral hole without disturbing the structure should not exceed 0.414 times that of the structure forming a sphere. | The sphere which can be placed in a tetrahedral hole without disturbing the close-packed structure should not have a radius larger than 0.225 times the radius of the sphere-forming the structure. |
| 5. | Radius of a tetrahedral void `"r"/"R"` = 0.225 | Radius of an octahedral void `"r"/"R"` = 0.414 |
shaalaa.com
Packing in Crystals
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
APPEARS IN
संबंधित प्रश्न
The ratio of close packed atoms to tetrahedral hole in cubic packing is ____________.
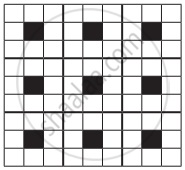
A two-dimensional solid pattern formed by two different atoms X and Y is shown below. The black and white squares represent atoms X and Y respectively. the simplest formula for the compound based on the unit cell from the pattern is
Distinguish between hexagonal close packing and cubic close packing.
Explain AAAA and ABABA and ABCABC type of three dimensional packing with the help of neat diagram.
Calculate the percentage efficiency of packing in the case of body centered cubic crystal.
What is the two dimensional coordination number of a molecule in square close packed layer?
