Advertisements
Advertisements
प्रश्न
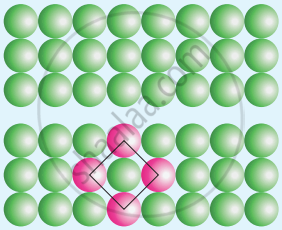
What is the two dimensional coordination number of a molecule in square close packed layer?
उत्तर
Square close packing: When the spheres of the second row are placed exactly above those of the first row. This way the spheres are aligned horizontally as well as vertically. The arrangement is AAA type. The coordination number is 4.
APPEARS IN
संबंधित प्रश्न
The ratio of close packed atoms to tetrahedral hole in cubic packing is ____________.
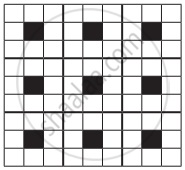
A two-dimensional solid pattern formed by two different atoms X and Y is shown below. The black and white squares represent atoms X and Y respectively. the simplest formula for the compound based on the unit cell from the pattern is
Distinguish between hexagonal close packing and cubic close packing.
Distinguish tetrahedral and octahedral voids.
Explain AAAA and ABABA and ABCABC type of three dimensional packing with the help of neat diagram.
Calculate the percentage efficiency of packing in the case of body centered cubic crystal.
