Advertisements
Advertisements
प्रश्न
Explain AAAA and ABABA and ABCABC type of three dimensional packing with the help of neat diagram.
उत्तर
1. AAAA type of three-dimensional packing: This type of three-dimensional packing arrangement can be obtained by repeating the AAAA type two-dimensional arrangements in three dimensions, i.e., spheres in one layer sitting directly on the top of those in the previous layer so that all layers are identical.
All spheres of different layers of crystal are perfectly aligned horizontally and also vertically so that any unit cell of such arrangement as simple cubic structure as shown in fig.

Simple Cubic (SC)
In simple cubic packing, each sphere is in contact with 6 neighbouring spheres – Four in its own layer, one above and one below and hence the coordination number of the sphere in a simple cubic arrangement is 6.
2. ABABA type of three-dimensional packing: In this arrangement, the spheres in the first layer (A-type) are slightly separated and the second layer is formed by arranging the spheres in the depressions between the spheres in layer A as shown in the figure.
The third layer is a repeat of the first. This pattern ABABAB is repeated throughout the crystal. In this arrangement, each sphere has a coordination number of 8, four neighbors in the layer above and four in the layer below.
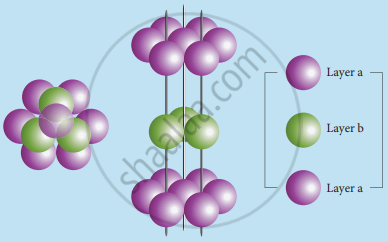
aba arrangement - hcp structure
3. ABCABC type of three-dimensional packing: In this arrangement (FCC) second layer spheres are arranged at the dips of the first layer. Third layer spheres are arranged in a manner such that they cover the octahedral void. Then no longer the third layer is similar to the first or second layer.
Third layer gives a different arrangement. Fourth layer spheres are similar to the first layer. If the first, second and third layers are represented as A, B, C then this type of packing gives the arrangement of layers as ABCABC… (i.e.,), the first three layers do not resemble the first, second and third layers respectively and the sequence is repeated.
with the addition of more layers. In this arrangement, atoms occupy 74% of the available space and thus has 26% vacant space. The coordination number is 12. Voids – The empty spaces between the three-dimensional layers are known as voids. There are two types of common voids possible. They are tetrahedral and octahedral voids.
Tetrahedral void – A void formed by three spheres of a layer in contact with each other and also with a sphere on the top or bottom layer is a hole between four spheres. The spheres are arranged at the vertices of a regular tetrahedron such as a hole or void is called a tetrahedral void.

abc arrangement – ccp structure
Octahedral void: A hole or void formed by three spheres of a hexagonal layer and another three spheres of the adjacent layer is a hole between six spheres. The spheres are arranged at the vertices of a regular octahedron. Such a hole or void is abc arrangement – ccp structure called octahedral void.
APPEARS IN
संबंधित प्रश्न
The ratio of close packed atoms to tetrahedral hole in cubic packing is ____________.
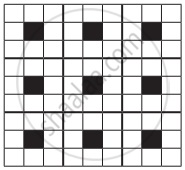
A two-dimensional solid pattern formed by two different atoms X and Y is shown below. The black and white squares represent atoms X and Y respectively. the simplest formula for the compound based on the unit cell from the pattern is
Distinguish between hexagonal close packing and cubic close packing.
Distinguish tetrahedral and octahedral voids.
Calculate the percentage efficiency of packing in the case of body centered cubic crystal.
What is the two dimensional coordination number of a molecule in square close packed layer?
