Advertisements
Advertisements
प्रश्न
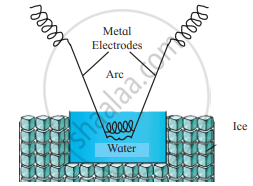
Draw labelled diagram of Bredig’s arc method.
उत्तर
APPEARS IN
संबंधित प्रश्न
Write Hardy-Sulze rules.
What happens when a beam of light is passed through a colloidal sol.
Explain Bredig’s arc method.
Draw labelled diagram Dialysis.
Froth and whipped cream are examples of ____________.
Identify the CORRECT statements from the following.
i. The colour of colloidal dispersion depends on size of colloidal particles.
ii. Tyndall effect is used to distinguish between colloidal dispersion and true solution.
iii. Eosin and congo red are examples of negatively charged sols.
In coagulation of positively charged ferric hydroxide sol, the most and least effective ions are ____________ and ____________ respectively.
Which of the following is an example of liquid-liquid system?
Maximum coagulation power is in ____________.
Mixing of two oppositely charged sols leads to ____________.
Froth is a colloidal solution of ____________.
The movement of colloidal particles towards respective electrode under electric field is called ______.
In which of the following sols there is low affinity between dispersed phase and dispersion medium?
Freshly prepared precipitate sometimes gets converted to colloidal solution by ______.
Which of the following process is responsible for the formation of delta at a place where rivers meet the sea?
What is collodion?
A colloid is formed by adding \[\ce{FeCl3}\] in excess of hot water. What will happen if excess sodium chloride is added to this colloid?
Why are some medicines more effective in the colloidal form?
How does the precipitation of colloidal smoke take place in Cottrell precipitator?
On the basis of Hardy-Schulze rule explain why the coagulating power of phosphate is higher than chloride.
Why does bleeding stop by rubbing moist alum?
For coagulating 200 ml of arsenious sulphide sol, 10 ml of 1 m NaCl solution is required find out the flocculation value of NaCl.
Which of the following is most powerful to coagulate the negative colloid?
Which of the following electrolytes will be most effective in coagulating negative sol?
Van Arkel's method of purification of metals involves converting the metal to a ______.
Identify positively charged sol from following.
