Advertisements
Advertisements
प्रश्न
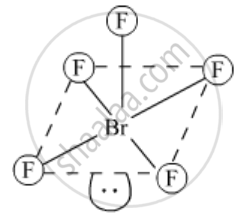
Draw the structures of the following: BrF5
उत्तर
BrF5
APPEARS IN
संबंधित प्रश्न
ICl is more reactive than I2.
Draw the structures of the following : ClF3
Draw the structure of IF7. Write its geometry and the type of hybridization.
Explain why:
(1) Interhalogen compounds are more reactive than the related elemental halogens.
(2) Sulphur exhibits the tendency for catenation but oxygen does not.
(3) On being slowly passed through water, PH3 forms bubbles but NH3 dissolves.
Which among the following pairs of halogen forms the interhalogen compound of the type \[\ce{XX^{'}_7}\]?
What is the oxidation state of bromine in the product?
\[\ce{Br2 + \underset{(excess)}{3F2} ->?}\]
The CORRECT statement about bromine trifluoride is:
Which among the following does not form polyhalide ion?
Which of the following exists in a yellow powder form at 25°C?
Which of the following interhalogen is a colourless liquid at room temperature?
