Advertisements
Advertisements
प्रश्न
Write the name of the cell which is generally used in transistors. Write the reactions taking place at the anode and the cathode of this cell.
उत्तर
The dry cell (also known as Leclanche cell) is used in transistors.
The reactions taking place at the anode and cathode are given below.
At anode: Zn(s) → Zn2+ + 2e−
At cathode : `MnO_2 + NH_4^+ + e^(-) -> MnO(OH) + NH_3`
APPEARS IN
संबंधित प्रश्न
On calculating the strength of current in amperes if a charge of 840C (coulomb) passes through an electrolyte in 7 minutes, it will be
- 1
- 2
- 3
- 4
96500 coulombs correspond to the charge on how many electrons?
Using the E° values of A and B, predict which is better for coating the surface of iron [E°(Fe+2/Fe) = -0.44V] to prevent corrosion and why?
Given: E° (A+2/A)=-2.37 V: E°(B+2/B)= -0.14V
How much charge is required for the following reduction:
1 mol of \[\ce{Cu^{2+}}\] to \[\ce{Cu}\]?
Three electrolytic cells A, B, C containing solutions of \[\ce{ZnSO4}\], \[\ce{AgNO3}\] and \[\ce{CuSO4}\], respectively, are connected in series. A steady current of 1.5 amperes was passed through them until 1.45 g of silver deposited at the cathode of cell B. How long did the current flow? What mass of copper and zinc were deposited?
What will happen during the electrolysis of aqueous solution of \[\ce{CuSO4}\] by using platinum electrodes?
(i) Copper will deposit at cathode.
(ii) Copper will deposit at anode.
(iii) Oxygen will be released at anode.
(iv) Copper will dissolve at anode.
Consider the figure and answer the following question.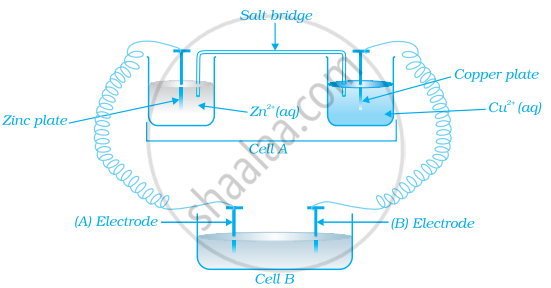
Cell ‘A’ has ECell = 2V and Cell ‘B’ has ECell = 1.1V which of the two cells ‘A’ or ‘B’ will act as an electrolytic cell. Which electrode reactions will occur in this cell?
Given `1/a` = 0.5 CM–1, R = 50 ohm, N = 1.0 then equivalent conductance of electrolytic cell is
On Electrolysis of dilute sulphuric acid using platinum electrodes, the product obtained at the anode will be.
The quantity of electricity needed to separately electrolyse 1 M solution of ZnSO4, AlCl3, and AgNO3 completely is in the ratio of ______.
