Advertisements
Advertisements
प्रश्न
Write the name of the cell which is generally used in transistors. Write the reactions taking place at the anode and the cathode of this cell.
उत्तर
The dry cell (also known as Leclanche cell) is used in transistors.
The reactions taking place at the anode and cathode are given below.
At anode: Zn(s) → Zn2+ + 2e−
At cathode : `MnO_2 + NH_4^+ + e^(-) -> MnO(OH) + NH_3`
APPEARS IN
संबंधित प्रश्न
Write any four applications of electrochemical series
Number of faradays of electricity required to liberate 12 g of hydrogen is:
Using the E° values of A and B, predict which is better for coating the surface of iron [E°(Fe+2/Fe) = -0.44V] to prevent corrosion and why?
Given: E° (A+2/A)=-2.37 V: E°(B+2/B)= -0.14V
Following reactions occur at cathode during the electrolysis of aqueous sodium chloride solution:
Na+(aq) + e− ⟶ Na (s) E0 = 2.71 V
H+(aq) + e− ⟶ `1/2` H2 (g) E0 = 0.00 V
On the basis of their standard reduction electrode potential (E0) values, which reaction is feasible at the cathode and why?
Suggest a list of metals that are extracted electrolytically.
How much charge is required for the following reduction:
1 mol of \[\ce{Al^{3+}}\] to \[\ce{Al}\]?
How much charge is required for the following reduction:
1 mol of \[\ce{Cu^{2+}}\] to \[\ce{Cu}\]?
Draw neat labelled diagram of electrolytic refining of blister copper
How much quantity of electricity in coulomb is required to deposit 1.346 × 10-3 kg of Ag in 3.5 minutes from AgNO3 solution?
( Given: Molar mass of Ag is 108 × 10-3 kg mol-1 )
Consider the figure and answer the following question.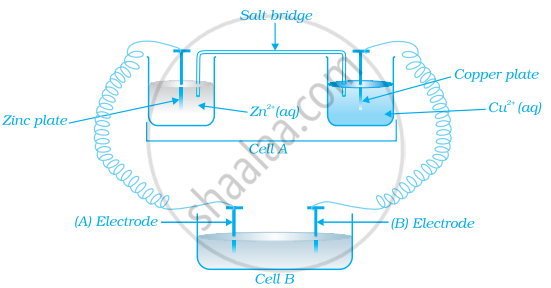
Cell ‘A’ has ECell = 2V and Cell ‘B’ has ECell = 1.1V which of the two cells ‘A’ or ‘B’ will act as an electrolytic cell. Which electrode reactions will occur in this cell?
