Advertisements
Advertisements
प्रश्न
Following reactions occur at cathode during the electrolysis of aqueous sodium chloride solution:
Na+(aq) + e− ⟶ Na (s) E0 = 2.71 V
H+(aq) + e− ⟶ `1/2` H2 (g) E0 = 0.00 V
On the basis of their standard reduction electrode potential (E0) values, which reaction is feasible at the cathode and why?
उत्तर
A redox reaction is feasible only if the species with high reduction potential is reduced (accepts an electron) and the species with lower standard reduction potential is oxidised (loses an electron). For a reaction to occur, it is necessary that the species with a greater tendency to lose an electron, loses its electron, which is gained by the species with a greater tendency to gain the electron.
The reactions taking place during the electrolysis of aqueous sodium chloride solution are
Na+ (aq)+e→ Na(s) E° =-2.71 V (I)
H+(aq)+ e → `1/2`H2(g) E° = 0.00 V (II)
One more reaction which takes place during the electrolysis of aqueous sodium chloride solution is
Cl2 +(aq)+2e → Cl (g) E° = 1.36 V (III)
From the above three reactions, we observe that the standard reduction potential sodium (reaction I) is negative, while the standard reduction potential for chlorine (reaction III) is positive. Hence, reaction III is feasible at the cathode.
APPEARS IN
संबंधित प्रश्न
The charge of how many coulomb is required to deposit 1.0 g of sodium metal (molar mass 23.0 g mol-1) from sodium ions is -
- 2098
- 96500
- 193000
- 4196
Number of faradays of electricity required to liberate 12 g of hydrogen is:
Using the E° values of A and B, predict which is better for coating the surface of iron [E°(Fe+2/Fe) = -0.44V] to prevent corrosion and why?
Given: E° (A+2/A)=-2.37 V: E°(B+2/B)= -0.14V
If a current of 0.5 ampere flows through a metallic wire for 2 hours, then how many electrons would flow through the wire?
How much charge is required for the following reduction:
1 mol of \[\ce{Al^{3+}}\] to \[\ce{Al}\]?
A solution of \[\ce{Ni(NO3)2}\] is electrolysed between platinum electrodes using a current of 5 amperes for 20 minutes. What mass of \[\ce{Ni}\] is deposited at the cathode?
Solve the following question.
A steady current of 2 amperes was passed through two electrolytic cells X and Y connected in series containing electrolytes FeSO4 and ZnSO4 until 2.8 g of Fe deposited at the cathode of cell X. How long did the current flow? Calculate the mass of Zn deposited at the cathode of cell Y.
(Molar mass : Fe = 56 g mol–1, Zn = 65.3 g mol–1, 1F = 96500 C mol–1)
Aqueous copper sulphate solution and aqueous silver nitrate solution are electrolysed by 1 ampere current for 10 minutes in separate electrolytic cells. Will the mass of copper and silver deposited on the cathode be same or different? Explain your answer.
Consider the figure and answer the following question.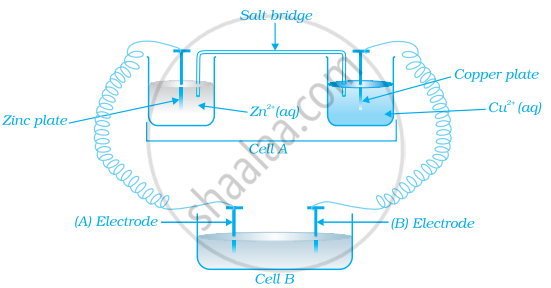
Cell ‘A’ has ECell = 2V and Cell ‘B’ has ECell = 1.1V which of the two cells ‘A’ or ‘B’ will act as an electrolytic cell. Which electrode reactions will occur in this cell?
When during electrolysis of a solution of Ag No3, 9650 coulombs of charge pass through the electroplating bath, the mass of silver deposite on the cathode will be:-
