Advertisements
Advertisements
प्रश्न
96500 coulombs correspond to the charge on how many electrons?
विकल्प
1.6 X 1019
6.022 X 1020
6.022 X 1023
6.022 X 1024
उत्तर
6.022 x 1023
APPEARS IN
संबंधित प्रश्न
Write any four applications of electrochemical series
State the first law of electrolysis
Number of faradays of electricity required to liberate 12 g of hydrogen is:
Using the E° values of A and B, predict which is better for coating the surface of iron [E°(Fe+2/Fe) = -0.44V] to prevent corrosion and why?
Given: E° (A+2/A)=-2.37 V: E°(B+2/B)= -0.14V
If a current of 0.5 ampere flows through a metallic wire for 2 hours, then how many electrons would flow through the wire?
How much charge is required for the following reduction:
1 mol of \[\ce{Al^{3+}}\] to \[\ce{Al}\]?
How much charge is required for the following reduction:
1 mol of \[\ce{Cu^{2+}}\] to \[\ce{Cu}\]?
Write any two uses of H2SO4
Draw neat labelled diagram of electrolytic refining of blister copper
Calculate the mass of Ag deposited at cathode when a current of 2 amperes was passed through a solution of AgNO3 for 15 minutes.
(Given : Molar mass of Ag = 108 g mol−1 lF = 96500 C mol−1)
How many faradays of electricity are required to produce 13 gram of aluminium from aluminium chloride solution? (Given: Molar mass of Al = 27.0-gram mol–1)
Following reactions occur at cathode during the electrolysis of aqueous copper(II) chloride solution :
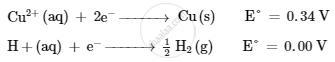
On the basis of their standard reduction electrode potential (E°) values, which reaction is feasible at the cathode and why ?
How much quantity of electricity in coulomb is required to deposit 1.346 × 10-3 kg of Ag in 3.5 minutes from AgNO3 solution?
( Given: Molar mass of Ag is 108 × 10-3 kg mol-1 )
Solve the following question.
A steady current of 2 amperes was passed through two electrolytic cells X and Y connected in series containing electrolytes FeSO4 and ZnSO4 until 2.8 g of Fe deposited at the cathode of cell X. How long did the current flow? Calculate the mass of Zn deposited at the cathode of cell Y.
(Molar mass : Fe = 56 g mol–1, Zn = 65.3 g mol–1, 1F = 96500 C mol–1)
Electrolytic cell uses electrical energy to bring about ____________.
According to Faraday’s First Law of Electrolysis, the amount of chemical reaction which occurs at any electrode during electrolysis by a current is proportional to the ____________.
What will happen during the electrolysis of aqueous solution of CuSO4 in the presence of Cu electrodes?
(i) Copper will deposit at cathode.
(ii) Copper will dissolve at anode.
(iii) Oxygen will be released at anode.
(iv) Copper will deposit at anode.
Assertion: Electrolysis of NaCl solution gives chlorine at anode instead of O2.
Reason: Formation of oxygen at anode requires overvoltage.
Consider the figure and answer the following question.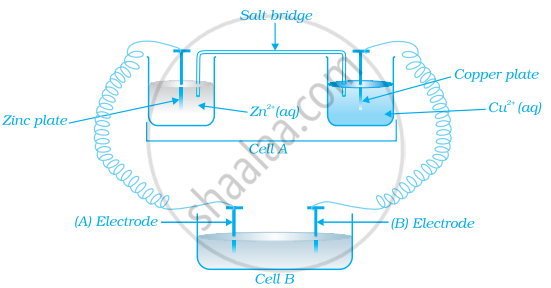
Cell ‘A’ has ECell = 2V and Cell ‘B’ has ECell = 1.1V which of the two cells ‘A’ or ‘B’ will act as an electrolytic cell. Which electrode reactions will occur in this cell?
When during electrolysis of a solution of Ag No3, 9650 coulombs of charge pass through the electroplating bath, the mass of silver deposite on the cathode will be:-
Given `1/a` = 0.5 CM–1, R = 50 ohm, N = 1.0 then equivalent conductance of electrolytic cell is
On Electrolysis of dilute sulphuric acid using platinum electrodes, the product obtained at the anode will be.
On passing electricity through nitrobenzene solution, it is converted into azobenzene. The mass of azobenzene is ______ mg, if the same quantity of electricity produces oxygen just sufficient to burn 96 mg of fullerene (C60)·
How much electricity in terms of Faraday is required to produce 40.0 g of \[\ce{Al}\] from molten \[\ce{Al2O3}\]?
(Given: Molar mass of Aluminium is 27 g mol−1.)
