Advertisements
Advertisements
प्रश्न
Following reactions occur at cathode during the electrolysis of aqueous copper(II) chloride solution :
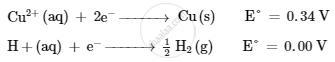
On the basis of their standard reduction electrode potential (E°) values, which reaction is feasible at the cathode and why ?
उत्तर
\[{Cu}^{2 +} \left( aq \right) + 2 e^- \to Cu\left( s \right) E^{\circ} = + 0 . 34 V\]
\[ H^+ \left( aq \right) + e^- \to \frac{1}{2} H_2 \left( g \right) E^{\circ} = 0 . 00 V\]
The relationship between the standard free energy change and the standard emf of a cell reaction is given by
Thus, the more positive the standard reduction potential of a reaction, the more negative is the standard free energy change associated with the process and, consequently, the higher is the feasibility of the reaction.
Since E∘Cu2+/CuECu2+/Cu° has a greater positive value than E∘H+/HEH+/H°, the reaction that is feasible at the cathode is
Cu2+(aq) + 2e− → Cu(s)
APPEARS IN
संबंधित प्रश्न
On calculating the strength of current in amperes if a charge of 840C (coulomb) passes through an electrolyte in 7 minutes, it will be
- 1
- 2
- 3
- 4
State the first law of electrolysis
Following reactions occur at cathode during the electrolysis of aqueous sodium chloride solution:
Na+(aq) + e− ⟶ Na (s) E0 = 2.71 V
H+(aq) + e− ⟶ `1/2` H2 (g) E0 = 0.00 V
On the basis of their standard reduction electrode potential (E0) values, which reaction is feasible at the cathode and why?
Draw neat labelled diagram of electrolytic refining of blister copper
How many faradays of electricity are required to produce 13 gram of aluminium from aluminium chloride solution? (Given: Molar mass of Al = 27.0-gram mol–1)
Electrolytic cell uses electrical energy to bring about ____________.
According to Faraday’s First Law of Electrolysis, the amount of chemical reaction which occurs at any electrode during electrolysis by a current is proportional to the ____________.
Assertion: Electrolysis of NaCl solution gives chlorine at anode instead of O2.
Reason: Formation of oxygen at anode requires overvoltage.
When during electrolysis of a solution of Ag No3, 9650 coulombs of charge pass through the electroplating bath, the mass of silver deposite on the cathode will be:-
Through an aqueous solution of an unknown salt of metal M (M = 200 g/mol) a current of 1.93 A is passed for 50 min. If 4 g of metal is produced at cathode. The charge on metal ion in solution is ______.
