Advertisements
Advertisements
प्रश्न
Following reactions occur at cathode during the electrolysis of aqueous copper(II) chloride solution :
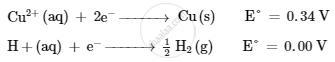
On the basis of their standard reduction electrode potential (E°) values, which reaction is feasible at the cathode and why ?
उत्तर
\[{Cu}^{2 +} \left( aq \right) + 2 e^- \to Cu\left( s \right) E^{\circ} = + 0 . 34 V\]
\[ H^+ \left( aq \right) + e^- \to \frac{1}{2} H_2 \left( g \right) E^{\circ} = 0 . 00 V\]
The relationship between the standard free energy change and the standard emf of a cell reaction is given by
Thus, the more positive the standard reduction potential of a reaction, the more negative is the standard free energy change associated with the process and, consequently, the higher is the feasibility of the reaction.
Since E∘Cu2+/CuECu2+/Cu° has a greater positive value than E∘H+/HEH+/H°, the reaction that is feasible at the cathode is
Cu2+(aq) + 2e− → Cu(s)
APPEARS IN
संबंधित प्रश्न
State the first law of electrolysis
How much charge is required for the following reduction:
1 mol of \[\ce{MnO^-_4}\] to \[\ce{Mn^{2+}}\]?
A solution of \[\ce{Ni(NO3)2}\] is electrolysed between platinum electrodes using a current of 5 amperes for 20 minutes. What mass of \[\ce{Ni}\] is deposited at the cathode?
Write any two uses of H2SO4
State second law of electrolysis
Calculate the mass of Ag deposited at cathode when a current of 2 amperes was passed through a solution of AgNO3 for 15 minutes.
(Given : Molar mass of Ag = 108 g mol−1 lF = 96500 C mol−1)
How much quantity of electricity in coulomb is required to deposit 1.346 × 10-3 kg of Ag in 3.5 minutes from AgNO3 solution?
( Given: Molar mass of Ag is 108 × 10-3 kg mol-1 )
In the electrolysis of aqueous sodium chloride solution which of the half cell reaction will occur at anode?
Given `1/a` = 0.5 CM–1, R = 50 ohm, N = 1.0 then equivalent conductance of electrolytic cell is
Through an aqueous solution of an unknown salt of metal M (M = 200 g/mol) a current of 1.93 A is passed for 50 min. If 4 g of metal is produced at cathode. The charge on metal ion in solution is ______.
