Advertisements
Advertisements
प्रश्न
Consider the figure and answer the following question.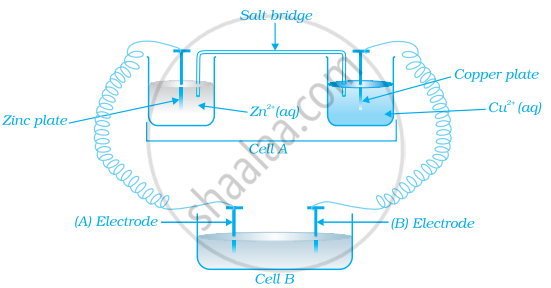
Cell ‘A’ has ECell = 2V and Cell ‘B’ has ECell = 1.1V which of the two cells ‘A’ or ‘B’ will act as an electrolytic cell. Which electrode reactions will occur in this cell?
उत्तर
Cell ‘B’ will act as electrolytic cell as it has lower emf
∴ The electrode reactions will be:
\[\ce{Zn^{2+} + 2e^{-} -> Zn}\] at cathode
\[\ce{Cu -> Cu^{2+} + 2e^{-}}\] at anode
APPEARS IN
संबंधित प्रश्न
96500 coulombs correspond to the charge on how many electrons?
Number of faradays of electricity required to liberate 12 g of hydrogen is:
How much charge is required for the following reduction:
1 mol of \[\ce{Cu^{2+}}\] to \[\ce{Cu}\]?
Three electrolytic cells A, B, C containing solutions of \[\ce{ZnSO4}\], \[\ce{AgNO3}\] and \[\ce{CuSO4}\], respectively, are connected in series. A steady current of 1.5 amperes was passed through them until 1.45 g of silver deposited at the cathode of cell B. How long did the current flow? What mass of copper and zinc were deposited?
Draw neat labelled diagram of electrolytic refining of blister copper
How many faradays of electricity are required to produce 13 gram of aluminium from aluminium chloride solution? (Given: Molar mass of Al = 27.0-gram mol–1)
In the electrolysis of aqueous sodium chloride solution which of the half cell reaction will occur at anode?
Time Required to deposite one millimole of aluminium metal by the passage of 9.65 ampere through aqueous solution of aluminium is
The quantity of electricity needed to separately electrolyse 1 M solution of ZnSO4, AlCl3, and AgNO3 completely is in the ratio of ______.
How much electricity in terms of Faraday is required to produce 40.0 g of \[\ce{Al}\] from molten \[\ce{Al2O3}\]?
(Given: Molar mass of Aluminium is 27 g mol−1.)
